Parkinson’s UK calls on government to mark World Patient Safety Day
PharmaTimes
SEPTEMBER 21, 2023
The report outlines ways the government can help patients with Parkinson’s disease - News - PharmaTimes
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

PharmaTimes
SEPTEMBER 21, 2023
The report outlines ways the government can help patients with Parkinson’s disease - News - PharmaTimes
Pharmaceutical Technology
JUNE 30, 2022
Pfizer and BioNTech have entered a new vaccine supply agreement worth $3.2bn with the US government to supply further doses of their Covid-19 vaccine. As per the agreement, the government will procure 105 million 3µg, 10 µg and 30 µg doses of the vaccine.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharma Marketing Network
MAY 3, 2023
In this article, we will explore the regulatory environment of pharmaceutical marketing and how companies are navigating it to balance patient safety and industry innovation. First, it is important to understand the different regulatory agencies that govern pharmaceutical marketing.

European Pharmaceutical Review
MARCH 12, 2024
Life science companies are increasingly using advanced technologies like artificial intelligence (AI), machine learning (ML) and blockchain to improve data quality and integrity by detecting anomalies in data and establishing tamper-proof audit trails for all data-related activities Data governance. How can this be mitigated?

European Pharmaceutical Review
MAY 2, 2024
It summarises the MHRA’s view on the UK government’s white paper on AI regulation published in 2023. Following publication of this white paper, the government requested that the MHRA outline the steps the agency is working on in relation to its expectations.

World of DTC Marketing
JANUARY 8, 2021
At the time, pharmaceutical giant Pfizer and its partner BioNTech—which now have the other US-authorized COVID-19 vaccine—had inked a deal with the US government to supply doses at $19.50 Also, Johnson & Johnson had a deal to supply the US government with doses of its vaccine—still in the works—at a rate of $10 per dose.
pharmaphorum
JANUARY 16, 2023
. “Although the totality of the data currently suggests that it is very unlikely that the signal…represents a true clinical risk, we believe it is important to share this information with the public, as we have in the past, when one of our safety monitoring systems detects a signal,” said the statement. 1 variant of Omicron.

World of DTC Marketing
AUGUST 26, 2021
The government has latitude to protect citizens from deadly conditions, especially when the science supporting vaccination is so clear” With respect to children, parents do not have carte blanche. That is a non-starter in the midst of a pandemic. The Constitution is not a suicide pact guaranteeing a right to harm others.
World of DTC Marketing
APRIL 15, 2021
Also cited was a lack of trust in the government to ensure the vaccines’ safety and effectiveness. And while there’s concern that the Covid-19 vaccines were rushed and that that means they haven’t been properly vetted or that their safety is otherwise in question, it’s simply not the case.

Pharmaceutical Technology
OCTOBER 17, 2022
The UK Government has announced funding worth more than $894.6m (£800m) to back research into ground-breaking, experimental therapies and progress the response of the country to patient safety challenges. These research centres comprise a new facility in Exeter.

pharmaphorum
NOVEMBER 28, 2022
According to a report by the Office of Health Economics (OHE), commissioned by the Association of the British Pharmaceutical Industry (ABPI), immediate action must be taken by governments, health systems, and companies to secure the era of green pharmaceuticals.
Pharmacy Times
JULY 25, 2022
Unvaccinated first responders were more likely to doubt the safety and efficacy of COVID-19 vaccines than vaccinated counterparts, reporting low trust in the government.

Nixon Gwilt Law
APRIL 22, 2024
Its three separate Rules (Security, Privacy, and Breach) impose a variety of security controls, documentation, governance, and notice requirements on custodians of protected health information (PHI). The HTI-1 Final Rule is an AI disclosure and governance rule published by the US Department of Health and Human Services (HHS).

Pharmaceutical Technology
DECEMBER 2, 2022
Under the Mission COVID Suraksha Program, DBT, the Indian Government and BIRAC provided funding for the development and clinical trials of the vaccine. Phase III trials of BBV154 were conducted to evaluate its safety and immunogenicity in approximately 3100 participants in 14 trial sites across the country.

PM360
OCTOBER 28, 2022
With this in mind, in order to maximize the benefits of AI while mitigating its related risks, pharmaceutical incumbents and startups need to implement AI governance processes and tools. Likewise, it enables them to predict the effect of the compound on the target and the compound’s safety risks. Promising AI Use Cases in Pharma.
Pharmaceutical Technology
AUGUST 15, 2022
After the vaccine was found to meet the standards of safety and quality, the regulatory agency granted the authorisation. According to the safety monitoring, side effects were generally mild and self-resolving, as well as in line with those reported for the original Moderna booster dose. 1 Omicron variant of concern.
Pharmaceutical Technology
APRIL 3, 2023
Under this system, patients can request the government for access to medical treatment using unapproved drugs not covered by insurance. Treatment is then conducted as a clinical trial by the National Cancer Center Hospital to confirm its safety and efficacy.

Pharmaceutical Technology
SEPTEMBER 5, 2022
These litigations were filed under the qui tam or whistleblower provisions of the False Claims Act, which facilitate private citizens to file lawsuits against fake claims on behalf of the government. Additionally, the company was accused of downplaying Trasylol’s safety risks.

Contrarian Sales Techniques
FEBRUARY 24, 2023
Key Initiatives for Public Healthcare, Education, And Infrastructure The Malaysian government just re-announced its Budget 2023 , highlighting various initiatives aimed at boosting the country's economy and providing support to its citizens. Economic growth and tax policies: The government predicts a 4.5% economic growth for the year.

World of DTC Marketing
OCTOBER 20, 2020
J&J’s refusal to release any information regarding a COVID-19 vaccination trial that J&J paused earlier this month comes on the heels of a recent and widely-touted joint pledge of prudence and caution on vaccine trial safety by nine drug companies, including J&J, working on COVID 19 vaccines. Make no mistake about it.

European Pharmaceutical Review
NOVEMBER 20, 2023
“This state-of-the-art parenteral site with the latest technology will enable us to continue to deliver medicines with safety first and quality always around the world,” stated Edgardo Hernandez, Executive Vice President and President of Eli Lilly’s manufacturing operations.

World of DTC Marketing
JULY 21, 2021
3hree: DTC marketing minimizes the side effects of prescription drugs – One of the top pages within prescription drug websites continues to be “safety information.” If drug companies didn’t do any DTC marketing, it would not affect drug prices. appeared first on World of DTC Marketing.com.

European Pharmaceutical Review
JUNE 16, 2023
The authors argued that fundamentally, implementation of AI/ML for pharmacovigilance has been hindered by the associated legal framework processes and governance issues. concluded: “If we do not harness the properties of data to allow us to leverage AI optimally, we will fail to do all we can for patient safety.” Bate et al.
Pharmaceutical Technology
NOVEMBER 22, 2022
Additionally, a contract with the ministry for the Japanese government to procure one million courses of Xocova has become effective with the approval. The latest development is based on the expected efficacy of the drug and the acceptability of its safety profile reported from a Phase II/III clinical trial.

Pharma Marketing Network
JULY 26, 2023
As new healthcare challenges arise and the pharmaceutical landscape evolves, the FDA may be allocating its resources to focus on other pressing issues, such as drug approvals, monitoring emerging health threats, and enhancing drug safety.
Pharmaceutical Technology
SEPTEMBER 7, 2022
Bharat Biotech carried out the product development linked to preclinical safety assessment, large-scale production scale-up, as well as the development of formulation and delivery device, including trials in humans.
European Pharmaceutical Review
OCTOBER 1, 2022
In order to carry out this work, the Council stated that governments must know which patient populations require supply and have accurate tissue and cell availability data to action this. When this is done, appropriate transplant supply needs can be met and adequate funding for tissue, cell and organ donation schemes can be identified.
European Pharmaceutical Review
JULY 12, 2023
SIIPL) and PATH, with funding from the UK government. MenFive ® has been developed through a 13-year collaboration between the world’s largest vaccine manufacturer Serum Institute of India Pvt. It is the only vaccine that prevents meningitis caused by meningococcal group X. This means it could potentially be used in other world regions.

PM360
JUNE 26, 2023
This includes ensuring access and affordability of their products, which can positively impact their environmental social governance (ESG) score and reputation. These partnerships can be with local governments, healthcare providers, local startups, and non-governmental organizations. In a shareholder meeting, Merck & Co.

European Pharmaceutical Review
DECEMBER 21, 2022
The Dutch government offers a prime example. Despite the tidal wave of renewed interest in psychedelic therapies, there is still much to be learned about them, including their precise mechanisms of action, their long- and short-term safety profiles, the durability of their effect, and their adverse effects—which can be severe.

European Pharmaceutical Review
NOVEMBER 15, 2022
“This paper highlights how the revised 2022 WHO Biosimilars Guideline provides Clarity, Consistency and Confidence in state-of-the-art regulatory science for biosimilar medicines based on… experience and over 1 million patient-treatment years of safety data in Europe alone,” explained Julie Maréchal-Jamil, Co-Chair of the Biosimilars Committee. “We
European Pharmaceutical Review
AUGUST 22, 2023
In July 2023, the US government body approved the only monoclonal antibody for protection of infants in their first RSV season. Two studies investigated the safety of the RSV vaccine Abrysvo. It is approved for use at 32 through to 36 weeks pregnancy gestation. US FDA granted approval of Abrysvo to Pfizer Inc. percent and by 76.5
Pharmaceutical Technology
JULY 7, 2022
Enhanced safety outcome was seen in non-hospitalised Covid-19 patients who are at increased risk of disease progression versus placebo. No deaths were reported in the treatment arm through 28 days compared to nine in the placebo arm. 2 subvariants.
Contrarian Sales Techniques
FEBRUARY 25, 2023
The film examines how Western pharmaceutical companies and governments have blocked access to low-cost AIDS drugs for the world's poorest people, particularly in Africa, where HIV/AIDS has been most devastating. Another challenge is ensuring the quality and safety of generic drugs.
Pharmaceutical Technology
AUGUST 4, 2022
Sinovac Biotech has reported that its Covid-19 vaccine, CoronaVac, obtained approval from the Health Bureau of the Government of the Hong Kong Special Administrative Region of the People's Republic of China for usage in children of the age six months to three years.
Pharmaceutical Technology
APRIL 28, 2023
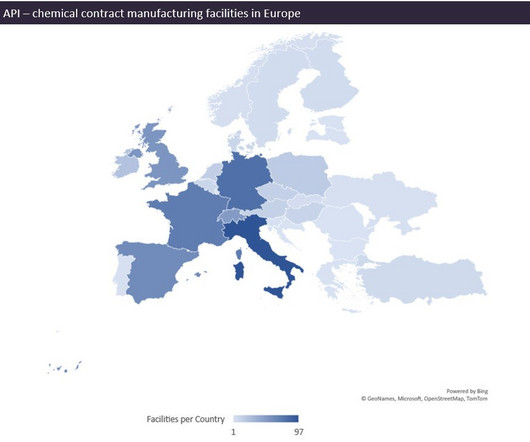
European stakeholders argue that, although the region may be more expensive than India or China, it brings supply security, quality, transportation speed, and more control for governments to steer production to critical pharmaceuticals. Europe also lacks suppliers to source key materials for API production.
European Pharmaceutical Review
AUGUST 3, 2022
In other vaccine news, the US Government has announced that it will provide more than $7 million to help expand access to COVID-19 vaccines in six African nations. is calling on specialists with experience in the formulation of mRNA vaccines and the analysis or QC of either finished products or their components to apply to join the party.
Pharmaceutical Technology
DECEMBER 22, 2022
Increased trafficking of illegal fentanyl represents a new threat to public health and safety. Fentanyl’s addictive properties mean that a) a consumer’s addiction will overpower any safety concerns they may have, and b) the drug can secure more addicts than it kills. A lethal drug that creates its own demand.

Clarivate
JUNE 29, 2023
Regulatory asymmetry: Laws governing generative AI vary widely across markets and are evolving rapidly as regulators scramble to address this emerging technology. Predicting safety and efficacy by using large language models to identify relevant documents and ways to optimize existing solutions.

World of DTC Marketing
AUGUST 20, 2020
It’s estimated that between 30-40% of people won’t get the vaccine because they either don’t believe in vaccines or they have lost trust in our government to approve a safe and effective vaccine. Polls show that as the pandemic has continued, US citizens have become less confident about the safety of a vaccine.
Contrarian Sales Techniques
FEBRUARY 26, 2023
These countries, known as "compulsory licensing" countries, have laws that allow the government to issue licenses to local companies to produce generic versions of patented drugs without the patent holder's permission. Some examples of compulsory licensing countries include India, Brazil, and South Africa.
Pharmaceutical Technology
SEPTEMBER 12, 2022
This dosage was chosen as the preferred dose in children aged under five years based on safety, tolerability and immunogenicity findings. In accordance with the recommendations of the Canadian government and public health authorities across the country, the companies plan to commence shipping the 3?g

European Pharmaceutical Review
NOVEMBER 1, 2023
However, now that researchers, investors and governments have seen how the vaccines created for COVID-19 have allowed large parts of the world to start moving again, it is vital we leverage this impetus to expedite the vaccines that the AMR crisis so badly needs. It is one of the leading causes of death associated with AMR globally.

Copyright Clearance Center
APRIL 4, 2023
Google recognizes that “drug approval” relates to “government regulations.” However, without integrating the company’s Adverse Event or Safety database, the search results could miss other unknown reactions such as itching, eczema, and hives. Compare this to another scenario. NIH’s MeSH [MeSH Browser, n.d.])
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content