2023 New Drug Approvals: Review of New FDA and EMA Marketing Authorisations
Tribeca Knowledge
NOVEMBER 28, 2023
T he drug-approval slump in major markets that clouded innovation prospects during 2022 seems to be over.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Tribeca Knowledge
NOVEMBER 28, 2023
T he drug-approval slump in major markets that clouded innovation prospects during 2022 seems to be over.
pharmaphorum
AUGUST 3, 2022
Immunotherapies and combination therapies have dramatically improved the prospects for newly-diagnosed SCLC patients, but despite these advances, many rapidly relapse due to the aggressive nature of the disease. The post Phase 3 lung cancer fail dents prospects of Ipsen, Servier’s Onivyde appeared first on.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
European Pharmaceutical Review
AUGUST 14, 2023
The US Food and Drug Administration (FDA) has approved Janssen’s Akeega (niraparib and abiraterone acetate), for the treatment of adult patients BRCA-positive metastatic castration-resistant prostate cancer (mCRPC). The FDA approval was based on positive results from the multi-centre Phase III MAGNITUDE study.

Pharma Leaders
MAY 18, 2023
The US Food and Drug Administration (FDA) has accepted Ardelyx’s resubmitted New Drug Application (NDA) for XPHOZAH (tenapanor) to control serum phosphate in adult patients with chronic kidney disease on dialysis who have had an insufficient response or intolerance to a phosphate binder therapy.

Legacy MEDSearch
MAY 24, 2023
iHealthScreen conducted a prospective trial in the general population to prospectively assess the product’s accuracy, sensitivity and specificity. These prospective study results were initially presented at the Annual Meeting of the American Academy of Ophthalmology (AAO) and selected for Panel Discussion as outstanding work.

Medico Reach
MARCH 24, 2023
The American Cancer Society estimates 1,958,310 new cases of cancer in the United States in 2023. So if you are a business looking to expand in the oncology market, read on to know more about oncology trends in 2023. billion in 2023 at a CAGR of 19.3%. The Cancer Immunotherapy Market Size is estimated to reach $152.6

Medico Reach
MARCH 24, 2023
The American Cancer Society estimates 1,958,310 new cases of cancer in the United States in 2023. So if you are a business looking to expand in the oncology market, read on to know more about oncology trends in 2023. billion in 2023 at a CAGR of 19.3%. The Cancer Immunotherapy Market Size is estimated to reach $152.6

pharmaphorum
JULY 6, 2022
The FDA has started an accelerated review of Eisai and Biogen’s a new amyloid-targeting antibody lecanemab for Alzheimer’s disease, with a 6 January deadline that keep them ahead of closest rival Eli Lilly. ” The post Eisai/Biogen could get FDA verdict on new Alzheimer’s drug by 6 Jan appeared first on. .
PM360
SEPTEMBER 21, 2023
Drug Approval ≠ CMS Coverage In April 2022, in advance of FDA approval of then-investigational monoclonal antibodies directed against amyloid treatments for AD, the Center for Medicare and Medicaid Services (CMS) issued a National Coverage Determination (NCD) related to all such amyloid treatments. Accessed August 10, 2023.

pharmaphorum
AUGUST 1, 2022
Sarepta is pressing forward with a bold plan to file with the FDA for accelerated approval of its gene therapy SRP-9001 for Duchenne muscular dystrophy (DMD) in the next few months, with a view to making it available in sometime around the middle of 2023.

Pharmaceutical Technology
MAY 22, 2023
Intercept Pharmaceuticals faced a setback after a US Food and Drug Administration’s (FDA) Advisory Committee (AdCom) meeting on the prospective use of its drug Ocaliva in pre-cirrhotic fibrosis due to nonalcoholic steatohepatitis (NASH) did not go in its favour.
pharmaphorum
DECEMBER 16, 2022
The positive opinion for etranacogene dezaparvovec – which was approved as Hemgenix by the FDA last month – raises the prospect of the first one-time therapy in the EU for the bleeding disorder, which affects around 1 in 50,000 of the population, according to the European Haemophilia Network (EUHANET).
European Pharmaceutical Review
JUNE 27, 2023
difficile in December 2022, the microbiome therapies space in 2023 has continued to see revolutionary developments and continued research advancement. Notably, gut microbiome was found to linked to cancer CAR T therapy response, based data from the largest prospective study of its kind, published in Nature Medicine in March 2023.

European Pharmaceutical Review
JULY 20, 2023
In June 2023, an array of leading voices in AAV development for cell and gene therapies articulated the current challenges and their optimism for the rapidly advancing sector at the Cell and Gene Therapy Summit event in London. More recently, the FDA granted approval of Roctavian in June 2023.
Pharmaceutical Technology
APRIL 19, 2023
The company is in discussions with industry experts and the National Institutes of Health (NIH) about the prospects of moving into Parkinson’s disease, said Kelleher-Andersson. The company has already started a Series A round to raise between $20 and $25 million, with a scheduled close in H2 2023, said Kelleher-Andersson.
European Pharmaceutical Review
DECEMBER 28, 2023
Finally, Andreas looks at the prospects for antibodies that target the innate immune system in treating both haematological malignancies and solid tumours. We received encouraging feedback from the US Food and Drug Administration (FDA) on the trial design and potential for accelerated approval.

pharmaphorum
NOVEMBER 21, 2022
With top-line results from the KALAHARI trial not due until the second half of 2023 – and cash reserves dwindling – the Belgian biotech hopes to look at the data after at least 25% of patients in the phase 2b portion of the study is completed. By the end of June, cash reserves had fallen to just over €3.7

pharmaphorum
JANUARY 29, 2023
One of the Johnson & Johnson’s top clinical trial readouts for 2023 just generated results, which bode very well for the future prospects of multiple myeloma CAR-T Carvykti.

Pharmaceutical Technology
MAY 9, 2023
Revenue prospects for Novo Nordisk’s blockbuster diabetes and weight-loss drug semaglutide in Brazil have been dealt a major blow after a federal court denied a request by the Danish firm for two patent extensions. Back in Brazil, however, the denial of semaglutide patent extensions is a loss for Lilly, too.

pharmaphorum
NOVEMBER 16, 2022
Bleak prospects, indeed. The specifics of an exciting prospect. This drug is not FDA or EMA approved, let’s be very clear, but it’s going to be – the PDUFA date for the FDA is 26th November […] If it is approved, I think the field feels very strongly that this is going to be a very substantial value-add to the field.

Legacy MEDSearch
JUNE 16, 2023
The prospective, randomized clinical trial is designed to evaluate the safety and effectiveness of the pro disc C Vivo and pro disc C SK system by comparing it with an approved TDR product as a control for 2-level indications, making it the first and only of its kind with two investigational devices and a TDR control.

pharmaphorum
SEPTEMBER 30, 2022
Sarepta Therapeutics has followed through on its promise to file for accelerated approval of its gene therapy SRP-9001 for Duchenne muscular dystrophy (DMD), as it aims for a launch in the middle of 2023. The post Sarepta files Duchenne muscular dystrophy gene therapy with FDA appeared first on. Photo by Markus Spiske on Unsplash.

Contrarian Sales Techniques
DECEMBER 14, 2022
Bright Prospects Indicates by Zacks Industry Rank The large-cap pharmaceutical industry, which is a part of the Medical sector, has a favorable Industry rank of A. J&J's outlook for the business remains positive, as it continues to expect its pharmaceutical business to grow at a high rate in 2023. in 2023. **The

Legacy MEDSearch
APRIL 21, 2023
a privately held company that is developing innovative therapies in endovascular embolization, is pleased to announce it has received FDA Premarket Approval (PMA) of its Lava® Liquid Embolic System (Lava® LES) for treatment of peripheral arterial hemorrhage. Bay Area-based BlackSwan Vascular, Inc., Are you hiring?

pharmaphorum
SEPTEMBER 13, 2022
The FDA has started a priority review of Chiesi ‘s velmanase alfa, an enzyme replacement therapy for lysosomal storage disease (LSD) alpha-mannosidosis, with a decision expected in the first half of 2023. The post FDA starts review of first drug for alpha-mannosidosis, from Chiesi appeared first on.
Legacy MEDSearch
JUNE 22, 2023
Food and Drug Administration (FDA) clearance to use the Topcon NW400 retinal camera with its EyeArt AI system to automatically detect diabetic retinopathy (DR), adding to the already-cleared usage with Canon CR-2 AF and Canon CR-2 Plus AF cameras. ” This latest FDA clearance for the EyeArt v2.2.0 sensitivity and 91.1%

Legacy MEDSearch
MAY 1, 2023
The results were featured in the late-breaking session ( LBA01-05 ) at the American Urological Association (AUA) 2023 Annual Meeting in Chicago on Sunday, April 30 th. Results from the trial are under review by the FDA. We continue to work with the FDA and look forward to introducing this innovative solution to physicians and patients.”

European Pharmaceutical Review
APRIL 30, 2024
Vikas Gupta (VG): In the years preceding the Covid pandemic, the ATMP s market, including both the innovators as well as the contract development and manufacturing organisations (CDMOs), was a bit too optimistic about its growth prospects as it was building significant manufacturing capacity focusing primarily on viral vectors.

Pharmaceutical Technology
MARCH 17, 2023
But while appealing, the prospects of developing a merged approach that combines both digital and pharmacological therapeutics remain challenging. In the US, a group of lawmakers recently reintroduced the “Access to Prescription Digital Therapeutics Act of 2023” in the US Senate.
Pharmaceutical Technology
JULY 4, 2022
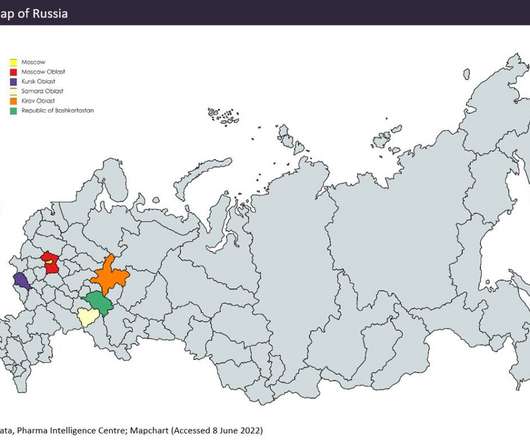
Compared with other emerging pharmaceutical markets (such as India and China, covered in previous Emerging Market Outsourcing Reports), Russian facilities lack many US Food and Drug Administration (FDA) and European Medicines Agency (EMA) approvals, meaning Russian manufacturing is more focused on the domestic market.

Evolve Your Success
APRIL 11, 2023
We have launched into the US marketplace and got the product design through FDA. In the second quarter of 2023, we said, “COVID moved past the time where it’s critical. My company standpoint is, “Online is the new prospecting tool.” Online is the new prospecting tool. I’ve got an FDA issue.”

Pharmaceutical Technology
JUNE 13, 2023
The US Food and Drug Administration (FDA) approved Ferring Pharmaceuticals’ Prior-Approval Supplement (PAS) to the Biologics License Application (BLA) for the bladder cancer gene-therapy Adstiladrin (nadofaragene firadenovec). Ferring plans to begin Adstiladrin production in the H2 2023, as part of an initial rollout in the US.
Pharmaceutical Technology
JANUARY 22, 2023
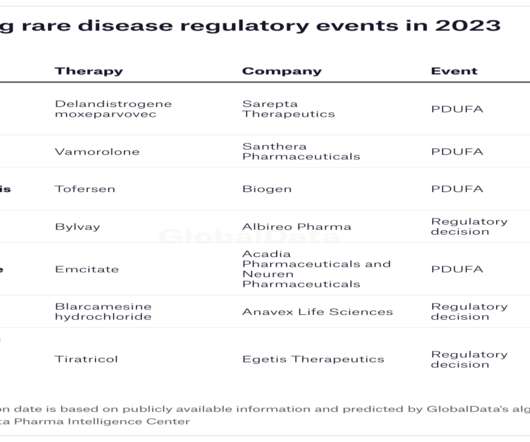
In 2022, the FDA approved only 37 new medicines, an underwhelming number compared to 98 in 2018. More therapies to meet unmet needs GlobalData predicts that at least 35 US regulatory decisions on drugs for rare diseases are on the horizon for 2023. GlobalData is the parent company of Pharmaceutical Technology.

Pharmaceutical Technology
MAY 24, 2023
Innoviva has snagged a win for its bacterial pneumonia drug Xacduro (durlobactam sodium + sulbactam) with a US Food and Drug Administration FDA approval for use in patients of ages 18 or older. The FDA approval stemmed from results from Entasis’ Phase III Attack trial (NCT03894046).
Pharmaceutical Technology
MAY 4, 2023
After a winter marked by a rise in hospitalisations due to the RSV, the FDA has granted its first approval for an Respiratory Syncytial Virus (RSV) vaccine to GSK’s Arexvy for adults ages 60 years and above. These recommendations and the FDA approval were based on positive AReSVi-006 Phase III trial data.

Pharmaceutical Technology
DECEMBER 4, 2022
There are seven FDA-approved Humira biosimilars, of which Amgen’s Amjevita will be launched in January. Indeed, Amgen’s biosimilar was approved in September 2016, but the company was not able to launch it until January 2023 in the US. While small molecule drugs can be remade into identical generics, biologics are more complex.

Pharmaceutical Technology
MARCH 21, 2023
FDA and EMA decisions In January, the European Commission (EC) approved Roche’s Xofluza (baloxavir marboxil) to prevent and treat influenza in children ages one year and older. Regulatory decisions by the FDA and EMA for select therapies from late January to late February and the CMOs contracted to manufacture the therapies.

Pharmaceutical Technology
APRIL 6, 2023
The US Food and Drug Administration (FDA) has released the fourth and final chapter in a series of guidance documents designed to support patient-focused drug development. The guidance also touched upon clinical trial design elements that must be explained, described, or rationalised to the FDA.
Pharmaceutical Technology
MAY 23, 2023
The US Food and Drug Administration (FDA) has granted Centessa Pharmaceuticals’ activated Protein C (APC) inhibitor SerpinPC a fast track designation for treating haemophilia B. The FDA’s fast track designation is designed to speed up the regulatory process to allow faster drug development and review to fill unmet needs.

Pharmaceutical Technology
MAY 15, 2023
If approved, the therapy could become the first FDA-approved gene transfer therapy for the condition. The FDA will assess the drug’s approval before its Prescription Drug User Fee Act (PDUFA) date on 29 May 2023. She said, “I wish the data were more persuasive”.

Pharmaceutical Technology
MAY 9, 2023
After the FDA rejected an accelerated approval bid in January, Eli Lilly has returned with topline data proving that donanemab may be an effective alternative to Eisai’s Leqembi (lecanemab) in treating Alzheimer’s disease. The FDA approved Biogen’s Aduhelm (aducanumab) for treating Alzheimer’s disease in June 2021.

Pharmaceutical Technology
MAY 9, 2023
After the FDA rejected an accelerated approval bid in January, Eli Lilly has returned with topline data proving that donanemab may be an effective alternative to Eisai’s Leqembi (lecanemab) in treating Alzheimer’s disease. The FDA approved Biogen’s Aduhelm (aducanumab) for treating Alzheimer’s disease in June 2021.

Pharmaceutical Technology
MAY 9, 2023
New York-based Eyenovia has announced an FDA approval for Mydcombi (tropicamide and phenylephrine hydrochloride), the first ophthalmic spray for mydriasis or pupil dilation. The company will provide further updates on Mydcombi and its ongoing development programs and partnerships during its scheduled Q1 2023 update conference call on May 11.

Pharmaceutical Technology
JUNE 2, 2023
While Leqembi was awarded an accelerated approval in January 2023, a decision on a traditional approval is expected by 6 July. The FDA will then consider the results of the committee for an approval decision in the following weeks. Clinical investigators commonly use registries to gather information on patient outcomes.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content