GSK's ViiV explores novel pathway to bring long-acting HIV med Cabenuva to tough-to-treat patients
Fierce Pharma
JULY 6, 2023
GSK’s Cabenuva made history in 2020 as the world’s firs | GSK's long-acting Cabenuva is currently approved only as a switch therapy for patients who already have undetectable HIV levels in the blood. Trying to reach some tough populations who have difficulty adhering to existing daily oral therapies, GSK is getting creative.







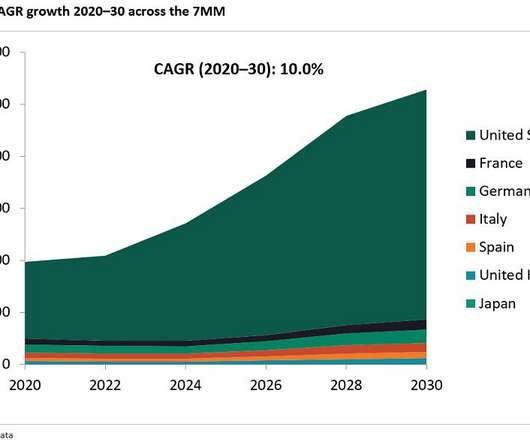





































Let's personalize your content