Millions of people to view digital prescriptions via the NHS app
PharmaTimes
FEBRUARY 1, 2024
Prescriptions ordered via the app are expected to save around 1.85 million hours in 2024
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

PharmaTimes
FEBRUARY 1, 2024
Prescriptions ordered via the app are expected to save around 1.85 million hours in 2024

MedCity News
FEBRUARY 4, 2024
2024 stands at a critical juncture in the battle against soaring pharmacy costs. In 2023, prescription drug costs rose by 8.4%, a 31% increase from the prior year. By enforcing transparency, PBMs would need to release detailed information on prescription drug spending. Transparency and reporting requirements.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Clarivate
APRIL 18, 2024
RxPass offers more than 50 select prescriptions at no additional cost for $5 per month. In classic Amazon style, its pharmacy offers two-day shipping on prescriptions by mail, with same-day delivery available in seven markets, with plans to add a dozen more cities by year end.

Healthcare Success
JANUARY 2, 2024
Now that the holidays have drawn to a close, it’s time to return to your 2024 marketing plan and make adjustments that ensure your business is taking advantage of the latest trends in healthcare marketing to reach patients and HCPs more effectively and make a lasting impression.

MedCity News
AUGUST 1, 2023
in 2024, down from $56.49 The average total monthly premium for Medicare Part D coverage is expected to be $55.50 in 2023, according to CMS.

MedCity News
JULY 12, 2023
CVS Caremark and GoodRx created a new program called Caremark Cost Saver, which will become available starting January 1, 2024. Through the program, eligible CVS Caremark commercially insured members will have access to GoodRx’s prescription pricing on generic medications.
MedCity News
AUGUST 8, 2023
Through Scripta Insights’ app, members of its customers can input the medications they’re taking, and the company will provide a savings report that will show different options on how they can save on their prescriptions. Starting in 2024, Cost Plus Drugs may show up as an option in the savings report.

PM360
FEBRUARY 8, 2024
Saatchi & Saatchi Wellness and TOM (Tikkun Olam Makers) The Prescription Paper Pill Bottle An innovative alternative to plastic, the Prescription Paper Pill Bottle is 100% compostable with no artificial glue and no toxic dye. It’s open-source design meets FDA requirements for safety and protection.

PharmExec
FEBRUARY 21, 2024
The FDA assigned the biologics license application for linvoseltamab to treat relapsed/refractory multiple myeloma with a Prescription Drug User Fee Act of August 22, 2024.

Pharmaceutical Commerce
FEBRUARY 20, 2024
The FDA has assigned Servier’s New Drug Application for vorasidenib to treat IDH-mutant gliomas with a Prescription Drug User Fee Act action date of August 20, 2024.

PharmExec
FEBRUARY 20, 2024
The FDA assigned the supplemental new drug application for Krazati (adagrasib) plus cetuximab in patients with locally advanced or metastatic colorectal cancer with a Prescription Drug User Fee Act goal date of June 21, 2024.
Pharmaceutical Commerce
JANUARY 24, 2024
The FDA assigned Autolus Therapeutics' biologics license application for obecabtagene autoleucel with a Prescription Drug User Fee Act date of November 16, 2024.
Pharmacy Times
MARCH 5, 2024
The continuous glucose monitoring system will be available for purchase online and without a prescription starting in summer 2024.

PharmExec
FEBRUARY 6, 2024
The FDA assigned a Prescription Drug User Fee Act action date of June 7, 2024, to an application that would expand the indication of Arexvy to include adults 50-59 years with an increased risk of respiratory syncytial virus-related lower respiratory tract disease.
Pharmaceutical Commerce
DECEMBER 19, 2023
Merck’s Biologics License Application for V116, a 21-valent pneumococcal conjugate vaccine, was given a Prescription Drug User Fee Act (PDUFA) date of June 17, 2024.
PharmExec
DECEMBER 19, 2023
Merck’s Biologics License Application for V116, a novel 21-valent pneumococcal conjugate vaccine has been given a Prescription Drug User Fee Act (PDUFA) date of June 17, 2024.
PharmExec
NOVEMBER 30, 2023
Supplemental new drug application for roflumilast cream 0.15% to treat atopic dermatitis in patients 6 years of age and older was assigned a Prescription Drug User Fee Act target action date of July 07, 2024.

PharmExec
FEBRUARY 20, 2024
The FDA set a Prescription Drug User Fee Act date during Q4 of 2024 for the biologics license application for datopotamab deruxtecan in patients with previously treated advanced nonsquamous non-small cell lung cancer.

Pharmaceutical Commerce
NOVEMBER 10, 2023
Blue Fin Group exec speaks on his upcoming feature.
Pharmacy Times
FEBRUARY 7, 2024
The FDA assigned a Prescription Drug User Fee Act (PDUFA) goal date of October 8, 2024.

Pharmacy Times
SEPTEMBER 29, 2023
The FDA has issued a Prescription Drug User Fee Act fate of March 26, 2024, for sotatercept (Merck) as treatment for adults with pulmonary arterial hypertension.

PM360
SEPTEMBER 21, 2023
As many in the industry now know, the Inflation Reduction Act (IRA) is the most significant reform of Medicare prescription drug coverage since the creation of Part D and has wide-ranging implications for the healthcare industry. CMS will present an initial offer for each drug’s maximum fair price (MFP) by February 1, 2024.

Pharmacy Times
NOVEMBER 7, 2023
AON’s specialty pharmacy reports success in reducing drug waste like unnecessary and early prescription refills.

Pharmaceutical Technology
MAY 19, 2023
The prescription drug user fee act (PDUFA) date is expected to be set as January 2024. The new investigational therapeutic product candidate STS101 is a nasal powder formulation of dihydroergotamine mesylate (DHE), an anti-migraine drug, which is given through the company’s nasal delivery device.

Legacy MEDSearch
MARCH 7, 2024
Dexcom disclosed its plans to make Stelo available for purchase online without the need for a prescription, starting in summer 2024. The device relies on a wearable sensor, which communicates with a smartphone application to continuously monitor blood glucose levels providing valuable insight into users’ blood sugar fluctuations.

Pharma Leaders
JUNE 8, 2023
Under PDUFA VII, the agency agreed to create the program by December 2022 and then to report aggregate data on an annual basis starting June 30, 2024, including data sources, study designs and the types of regulatory requests.
Pharmaceutical Technology
JUNE 15, 2023
The regulator has set 13 February 2024 as a Prescription Drug User Fee Act goal date for the review of Ipsen’s sNDA. The Onivyde regimen comprises Onivyde (irinotecan liposome injection) along with 5-fluorouracil/leucovorin and oxaliplatin (NALIRIFOX regimen).
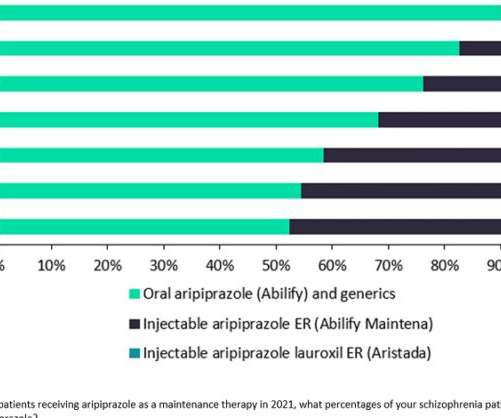
Pharmaceutical Technology
JULY 28, 2022
Prescription of daily oral aripiprazole continues to dominate the use of Otsuka and Lundbeck’s once-monthly LAI formulation Abilify Maintena and Alkermes’ Aristada, which can be administered every six weeks. GlobalData forecasts that it will launch in the US and 5EU next year.

pharmaphorum
NOVEMBER 2, 2022
Around 70% of new prescriptions for the drug are for patients who haven’t received this type of therapy in the past, with fewer than 10% switching from Trulicity, said Lilly’s chief financial officer Anat Ashkenazi on the call.

PM360
NOVEMBER 14, 2023
This collaboration builds on Sanofi’s recent announcement to lower the list price for Lantus and cap OOP costs at $35 for all patients with commercial insurance, which goes into effect January 1, 2024. The price is available to cash-paying patients with a valid prescription. Currently, an estimated 37.3 of the U.S.

Eversana Intouch
MARCH 19, 2024
That’s why in 2024, we find ourselves still failing women in healthcare. They are more likely than men to suffer chronic conditions and use prescription drugs across all age groups. When it comes to healthcare, however, this model often fails to consider the good of all. Women represent more of the health care system than men.

pharmaphorum
OCTOBER 3, 2022
The third ALS treatment to be given FDA approval, following Mitsubishi Tanabe’s Radicava ($170,000 per year) and generic drug riluzole – Relyvrio is priced at around $12,500 per 28-day prescription. Clearer’ data isn’t expected until 2024, with results from a late-stage phase 3 PHOENIX trial of around 600 patients due at that time.

Clarivate
OCTOBER 30, 2022
Of surveyed MCOs that cover or plan to cover DTx apps for the three conditions, more than 80% of respondents reported reimbursing DTx for at least one of the conditions in their fully insured commercial plans as of March 2022, with the remainder expecting to do so by 2024. Payers also noted some coverage in their Medicare and Medicaid plans.

Nixon Gwilt Law
JANUARY 16, 2024
What can digital health innovators and investors anticipate for the industry in 2024? What can digital health innovators and investors anticipate for the industry in 2024? Read on to find out what else is on our Legal Vision Board for 2024! How is 2024 Shaping up for Your Business?

PM360
NOVEMBER 13, 2023
When it comes to platforms used by HCPs to better understand prescription drug pricing information, GoodRx is second only to a provider’s electronic health record (EHR). Is GoodRx in Your 2024 HCP Marketing Plan? Click here to learn more about GoodRx HCP media solutions and give us a try in your 2024 HCP marketing plan.
pharmaphorum
SEPTEMBER 30, 2022
In March, the first advisory committee agreed with the opinion voiced by the FDA reviewer that there were doubts about the efficacy of Relyvrio, and that it would be preferable to wait for the results of an ongoing, larger study – now expected to read out in 2024.

PM360
APRIL 7, 2023
This is especially significant for manufacturers of high-cost specialty drugs, who will now face a recurring additional 20% charge on virtually every prescription fill. On the other side, drug manufacturers are facing the squeeze, with caps on price inflation and a mandated 20% discount in the catastrophic phase.

PM360
SEPTEMBER 21, 2023
This product would be a more potent prescription version than the OTCs, Casberg notes. In 2024, ARPA removes the 100% AMP cap. The company says it will produce two million doses a year and give away half of them for free, while the rest will be sold at roughly $18 per dose.

Clarivate
DECEMBER 19, 2022
Insurers that incentivize using Walmart facilities in Medicare Advantage and prescription drug plans (PDPs) have seen success; Humana has a long-running standalone Part D plan with Walmart pharmacies as preferred network locations.

MedCity News
MARCH 7, 2024
During a fireside chat hosted by MedCity News at ViVE, Blue Shield of California CEO Paul Markovich discussed the company’s decision to largely move away from CVS Caremark as its PBM and partner with several other companies.
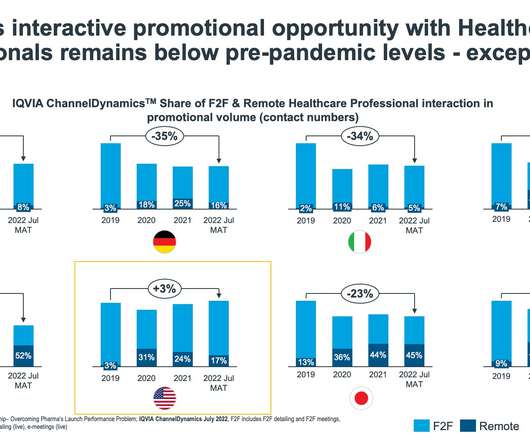
pharmaphorum
JANUARY 24, 2023
So far, only eight of the top 100 pharmaceutical companies by global prescription medicines sales have made a net zero commitment in the same date range. 2023 will therefore be a year of challenge and shakeout: what emerges to enter 2024 will be only the most innovative, agile, and above all, resilient.
PM360
SEPTEMBER 28, 2022
In April 2022, we released an article describing proposed policies from the federal government that could impact prescription drug pricing. How drug manufacturers should prepare for 2024: Have a strategy for request-for-proposal (RFP) season: RFPs for plan year 2024 will come out in late 2022 and run into early 2023.
European Pharmaceutical Review
JANUARY 5, 2023
Triomphe added: “… it is our intention to make nirsevimab available, if approved in time, for the 2023/2024 season to help alleviate the burden of RSV on families and the healthcare system.”. The Prescription Drug User Fee Act date, the FDA target action date for their decision, is in the third quarter of 2023.

Pharmaceutical Technology
JANUARY 6, 2023
The regulatory body has assigned a Prescription Drug User Fee Act (PDUFA) date for its decision in the third quarter of this year. If approved, nirsevimab will become the first single-dose preventative against RSV lower respiratory tract disease for the 2023/2024 RSV season in the US.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content