JPM23, Day 2: Takeda readies for dengue vaccine launch; Merck touts recent deals
Fierce Pharma
JANUARY 10, 2023
JPM23, Day 2: Takeda readies for dengue vaccine launch; Merck touts recent deals. Tue, 01/10/2023 - 06:24.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 launch-readiness
launch-readiness 
Fierce Pharma
JANUARY 10, 2023
JPM23, Day 2: Takeda readies for dengue vaccine launch; Merck touts recent deals. Tue, 01/10/2023 - 06:24.

Tribeca Knowledge
FEBRUARY 9, 2023
I n our latest report, Launch Readiness 2023, we look at the prospects and challenges for new medicines entering the market in the shadow of COVID-19. We explain how digital transformation and agile launch software can help companies track, manage and align their launch activities in real-time, across functions and markets.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Tribeca Knowledge
FEBRUARY 13, 2024
I n our latest report, Launch Readiness 2024 , we look at the prospects and obstacles facing new medicines in markets still recovering from the COVID-19 pandemic.

Fierce Pharma
FEBRUARY 3, 2023
Vaccines slip, Dupixent climbs for Sanofi as it readies multiple launches in 2023 zbecker Fri, 02/03/2023 - 22:02
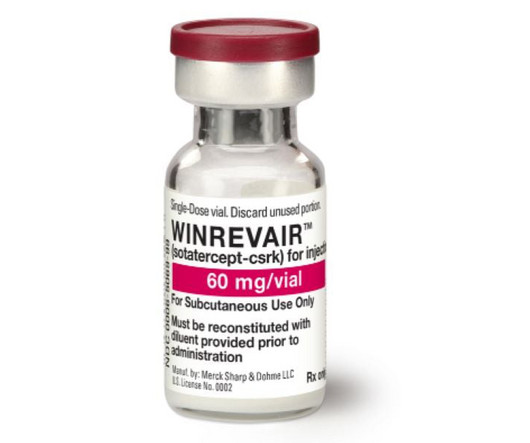
Fierce Pharma
MARCH 26, 2024
billion acquisition of Acceleron is ready to pay dividends. With the FDA’s approval of Winrevair (sotatercept) to treat pulmonary arterial hypertension (PAH), Merck is set to launch the first disease-modifying treatment for the condition. After three years, the crown jewel of Merck’s $11.5

Fierce Pharma
FEBRUARY 15, 2024
In the U.S., Alvotech’s biosimilar to Johnson & Johnson's blockbuster immunology drug Stelara is set to face market competition from the likes of

Tribeca Knowledge
FEBRUARY 1, 2022
I n our new report Launch Readiness 2022 , we consider how launch strategists may best navigate a fast-changing yet still commercially ripe environment for pharmaceutical launch readiness.

Tribeca Knowledge
FEBRUARY 24, 2021
I t is probably reasonable enough by now to talk about the launch readiness landscape for medicines and vaccines as pre- and post-COVID. We can also say with some certainty that nothing will ever be the same again.

PharmExec
JANUARY 19, 2023
Launch Ready from Day One is designed to empower a company's customer facing teams to drive demand, simplify access, and create a seamless and more integrated customer experience.

PharmaTech
APRIL 16, 2024
WMFTS has launched WMArchitect, a single-use product line that offers ready-to-use single-use assemblies and custom-designed workflows for biopharma fluid management.

Tribeca Knowledge
DECEMBER 8, 2022
As we map out in our pharma trends for 2023, COVID-related disruption has created a whole new set of launch readiness and market access challenges for companies looking to maximise their assets in the new year. T he pharmaceutical industry has little room for complacency in a post-COVID world.

Pharmaceutical Technology
OCTOBER 20, 2023
The company has said that it is ready to launch a Phase IIa trial with the drug being intended to treat an often painful condition.

World of DTC Marketing
DECEMBER 9, 2021
The days of enormous product launch budgets are over unless a new mass-market product is introduced. One top-10 pharma company has created a “market launch metric” team to measure key launch milestones. ” The post Do you have enough money to launch your drug?

World of DTC Marketing
APRIL 19, 2022
Before most drugs are launched, much time and effort are spent developing marketing plans and getting approval. Brand managers usually spend a lot of meetings getting approval for product launches, but that could be a huge mistake. You’re on top of trends that will affect your brand’s launch or growth in its life cycle.

PM360
NOVEMBER 12, 2023
Here’s the fact: since the pandemic, launch revenues in pharma have declined. The question is: What do you do to maximize the success of your launch? In my experience, enormous respect must be paid to the pre-launch market research that assesses the impact your brand will make on the market. Those insights are irreplaceable.
Pharmaceutical Technology
JUNE 15, 2023
The US Food and Drug Administration (FDA) has approved Amneal Pharmaceuticals’ 505(b)(2) new drug application (NDA) for a ready-to-use oncology injectable, PEMRYDI RTU. Amneal Pharmaceuticals claimed that PEMRYDI RTU is the first and only ready-to-use presentation of pemetrexed for injection.

European Pharmaceutical Review
MAY 23, 2023
“Emerging biotech companies responsible for a record 65 percent of the molecules in the R&D pipeline in 2022” made common infrastructure choices to prepare for a European launch, stated Isma Hachi, Director Emerging Biopharma for IQVIA.

World of DTC Marketing
JULY 30, 2021
When I worked on the launch of Cialis, our digital agencies were always available via IM. If you have a good agency, you should be sharing it with your colleagues and promoting them within your company so that when a new brand is ready to launch, selecting an agency is 90% done. This was a huge benefit for us.

Contrarian Sales Techniques
DECEMBER 20, 2023
A data alchemist, transforming numbers into dazzling displays and irresistible product launches. Here, strategic plans waltz with data analysis, while product launches pirouette under the spotlight of innovative thinking. So, are you ready to swap spreadsheets for swatches, data reports for dazzling displays?

World of DTC Marketing
AUGUST 13, 2020
While most offices remain closed pharma is preparing for the launch of new products and has to have the infrastructure to ensure a successful launch. Obviously the experience can vary wildly between companies but it’s a good time if you want to learn and are ready to challenge the status quo.

World of DTC Marketing
SEPTEMBER 30, 2020
HERE”S THE DEAL: Determining how much to spend for the launch of a new drug is a challenge we all face and you have to be ready to shift funds from channels that are failing to provide the lift in brand awareness to channels that hit the bullseye on your objectives. The post Launching a new drug: How much to spend on DTC?
PM360
NOVEMBER 29, 2022
As healthcare marketers, we often focus on the official launch of a new product as a pinnacle moment for its commercial success. But in many cases, success depends on actions taken before the launch date—specifically, how well the market has been primed to receive a novel product. Avoid overly promotional messaging.

PM360
APRIL 22, 2024
Understanding the “why” before the “how” as Med Affairs charts the course for pre- and post-launch strategies. With a focus on patient-centricity, data-driven insights, and collaborative partnerships across the healthcare ecosystem, it’s redefining the playbook for market shaping and building advocacy for a product pre- and post-launch.

Medico Reach
JULY 3, 2022
With so much at stake, all stakeholders must work cooperatively to ensure the product launch is a success. In that case, an effective product launch plan will help it overcome most of the market’s obstacles. The post will outline the successful steps for medical device product launch. Establish Launch Objectives.

Medical Device Success
JULY 4, 2021
The right evidence powers you through your regulatory process and aligns that process and your claims with your value propositions across stakeholders and then on through your product launch. TTi Health Research & Economics Market Readiness Assessment link. TTi Health Research & Economics Market Readiness Assessment link.

European Pharmaceutical Review
FEBRUARY 7, 2024
The site will launch the company’ s cell therapy platforms in the US for critical cancer trials and future commercial supply. AstraZeneca has announced a $300 million investment in a US-based manufacturing facility in Rockville, Maryland, which will focus initially on T-cell therapies for oncology indications.

Clarify Health
FEBRUARY 7, 2023
They were looking for ways to launch high-performing provider networks more efficiently and needed to quickly assess provider performance in 12 new markets. Using Clarify Networks, a cloud-based software that optimizes networks for market success , this organization was ready to enter all 12 new markets in just 3 months.
Pharmaceutical Technology
JUNE 21, 2023
Ketarx will be marketed under the FDA-approved label of ketamine and in dosages that include 10mg/ml, 50mg/ml and 100mg/ml, with the option to boost the concentration and ready-to-administer applications. Ketarx will then be commercially launched in the second quarter. Ketamine is currently on the FDA’s drug shortage list.

World of DTC Marketing
JANUARY 4, 2021
DTC marketers need to be ready to understand and help patients who request new medications via telehealth or email. They should not rely on a “digital team” who meets with the brand to launch digital initiatives. Become the “go-to” health site for new health information on the condition in which you compete.

PM360
JUNE 29, 2022
For example, if you are within a year of launch, embarking on a bold, novel launch strategy is likely not going to provide you with enough time to course correct if challenges arise. If you are not ready today, then determine how you will get ready for tomorrow. Use this to your advantage. Honesty is the best policy here.

European Pharmaceutical Review
DECEMBER 20, 2023
Though extensions are possible, extension criteria could be challenging to achieve in practice (eg, pan-EU launch within two years, proof of unmet need). The first is removal of the RDP extension for pan-EU launch within two years, instead requiring manufacturers to submit to local P&MA processes upon request by the member state.

European Pharmaceutical Review
OCTOBER 4, 2022
Results from the pilot are anticipated to be ready in three to four years, when a report will be issued and a workshop with relevant stakeholders will discuss the lessons learned and what support measures or regulatory changes should be made going forward.

World of DTC Marketing
MAY 3, 2022
2wo: The competition’s MS drugs were better – The MS market is lucrative, and competitors launched products that were better than Biogen’s with fewer side effects discussed by the MS community online. A new CEO will be appointed and will undoubtedly be charged with getting the company ready for sale.

Pharmacy Times
JUNE 14, 2023
- Adds first ready-to-use presentation of key injectable for treating non-squamous non-small cell lung cancer and malignant pleural mesothelioma. Represents a high value, complex product launch for Amneal’s expanding injectable portfolio.

Pharmaceutical Technology
JUNE 8, 2023
Our strong manufacturing capability and scale, including from our vaccine manufacturing site in Belgium, means we are ready to deliver the vaccine as countries begin to launch.” The marketing authorisation from the EC was supported by data obtained from a pivotal Phase III AReSVi-006 (adult respiratory syncytial virus) trial.

Legacy MEDSearch
NOVEMBER 22, 2022
The company expects to launch the VisionPort System in the US before the end of 2022. This is the first time that I have launched a product that adds value across all stakeholders, clinical and financial alike in such a profound way. New View Surgical, Inc., For more information, please visit www.newviewsurg.com. Closes $12.1M

Pharmaceutical Technology
NOVEMBER 8, 2022
Following the acquisition, Viatris will combine its research and development (R&D), international commercial footprint and regulatory expertise and supply chain with the deep expertise of Oyster Point in the ophthalmology space, as well as Phase III-ready pipeline of Famy Life Sciences.

Nixon Gwilt Law
JANUARY 23, 2023
Are you getting ready to launch an RTM offering or wondering if you should add a CCM or PCM service line to your existing revenue model? Click here to download the Medicare Non-Facility Reimbursement Chart for Care Management Services.

Contrarian Sales Techniques
DECEMBER 20, 2023
So, are you ready to trade the lab coat for a sales blazer? Are you ready to unleash your scientific spirit and become a healthcare champion in Ipoh? With dedication and your science-fueled drive, you can climb the ranks, shape the future of healthcare in Ipoh, and become a legend in the medical sales scene. Good luck!
Pharmaceutical Technology
APRIL 21, 2023
Our distribution partners have Cuvrior stocked and ready for immediate use. At Orphalan, our focus is on providing targeted solutions for rare diseases, as demonstrated by the successful launch of Cuprior in Europe.”

Contrarian Sales Techniques
DECEMBER 19, 2023
So, are you ready to ditch the textbooks and grab the scientific baton? Are you ready to trade the lab bench for the sales stage and ignite your career with purpose and passion? If the answer is a resounding "Yes!", Good luck!

European Pharmaceutical Review
NOVEMBER 28, 2022
FACILITATE , (FrAmework for ClInicaL trIal participants’ daTA reutilisation for a fully Transparent and Ethical ecosystem) is a patient-driven legal framework that intends to help return clinical trial data to study participants, ready for re-use in further research or healthcare practice.

Clarivate
DECEMBER 9, 2022
To help you get ready, we created a simple checklist of five steps you can take to prepare. Before the Unitary Patent goes live: The UPC announced a three-month sunrise period will precede the launch of the UP and UPC. Clarivate stands ready to help you navigate the new Unitary Patent system. Monitor UPC case law.

PM360
MAY 4, 2023
We are laser-focused on launching new, innovative solutions to further bring our vision to life,” said Rotsart. The recognition has further ignited the award-winner’s readiness and commitment to doing more. Operations at Adheris Health.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content