Building a Robust Provider Network with SDoH Insights
Clarify Health
OCTOBER 10, 2023
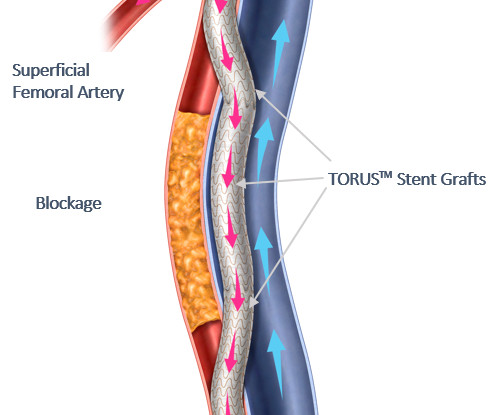
When building provider networks, it is crucial to include providers who can assess and address social determinants to ensure that care is equitable, patient-centered, and effective in improving health outcomes and reducing healthcare disparities.









































Let's personalize your content