FDA starts speedy review of Takeda’s dengue vaccine
pharmaphorum
NOVEMBER 23, 2022
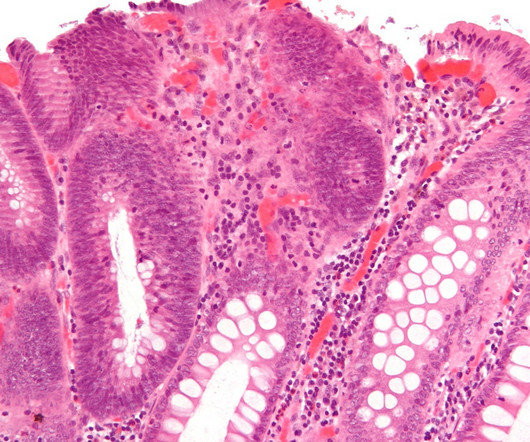
The Japanese pharma said it has filed for approval of TAK-003 with the FDA for the prevention of dengue disease caused by any dengue virus serotype in individuals aged four through 60. Dengue fever is unusual in that a first infection is rarely serious, but a second can lead to much more serious disease. billion in peak sales.





























Let's personalize your content