Study finds trials of prescription DTx often lack ‘rigour’
pharmaphorum
NOVEMBER 16, 2023
Study finds trials of prescription DTx often lack ‘rigour’ Phil.Taylor Thu, 16/11/2023 - 11:23 Bookmark this
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

pharmaphorum
NOVEMBER 16, 2023
Study finds trials of prescription DTx often lack ‘rigour’ Phil.Taylor Thu, 16/11/2023 - 11:23 Bookmark this

pharmaphorum
JANUARY 6, 2023
In 2023, expect manufacturers to further shift their focus away from acquiring new patient prescriptions to retaining existing customers. The account carried an official blue check mark, but in fact it did not belong to the prescription drugmaker. in 1979 to 2.8%in Ensuring patient access.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PM360
NOVEMBER 12, 2023
Technological innovations and changes in consumer behavior over the past 10 years have reduced friction, saved time, and opened new options for consumers and patients seeking routine prescriptions and refills via telehealth. Let’s face it, most consumers seek convenience wherever they can find it.

Fierce Pharma
JULY 21, 2023
In this episode of 'The Top Line' we replay an important show from April 28, 2023 regarding Narcan. | This week on "The Top Line," we revisit an episode on Narcan the nasal spray. It is the first naloxone product to be approved for use without a prescription.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
Ben Locwin will discuss the potential effects of this draft, along with the pharmacological and toxicological considerations for optimizing doses of prescription drugs. Explore the limitations of what we know about prescription drug dosing The future of Clinical Research April 12, 2023 at 11:00 am PDT, 2:00 pm EDT, 7:00 pm GMT

European Pharmaceutical Review
SEPTEMBER 4, 2023
Included in Issue 4 of European Pharmaceutical Review : FOREWORD Dissolution testing – a dual role David Elder, David P Elder Consultancy REGULATORY INSIGHT EU packaging reform: a prescription for change?
Pharmacy Times
JULY 28, 2023
Thousands more people filled their insulin prescriptions following the introduction of the cap in January 2023, especially compared to those who were not enrolled in Medicare, a new study finds.

Pharmaceutical Technology
MAY 30, 2023
According to GlobalData’s Looking Ahead to 2023 – the Future of Pharma report, five drugs set for approval in 2023 are projected to attain blockbuster status or near-blockbuster status by 2028 with US company dominance. These drugs are set to make a combined $4.34bn in sales in 2028. Similarly, Almirall S.A.,
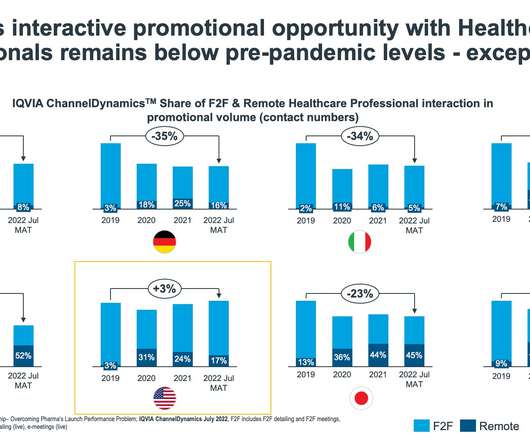
pharmaphorum
JANUARY 24, 2023
In this final instalment of IQVIA EMEA Thought Leadership’s Nine for 2023 three-part series, focusing on issues that will change the direction of healthcare and the pharmaceutical industry this year, three key competitive issues for pharmaceutical companies in 2023 are assessed.

Pharma Marketing Network
OCTOBER 26, 2022
Join Reuters Events’ exclusive panel discussion to hear experts from Novartis, Takeda and Janssen share business-critical learnings, including: Differentiate predictive vs prescriptive insights and inform your next best action. The post Pharma USA 2023 | March 28–29, 2023 appeared first on Pharma Marketing Network.

PharmExec
NOVEMBER 7, 2023
Exagamglogene autotemcel (Exa-Cel) was assigned a Prescription Drug User Fee Act action date of December 8, 2023, for the treatment of sickle cell disease.

PM360
SEPTEMBER 21, 2023
Jeff Casberg, MS, RPh, Senior Vice President of Clinical Pharmacy at IPD Analytics, a managed care and pharmaceutical consultancy, highlighted a wide range of 2023 key events that will affect managed care professionals by the end of the year. This product is expected to have three generic launches in 2023.

pharmaphorum
DECEMBER 23, 2022
And in 2023, these investments will hopefully pay off. In 2023, omnichannel behaviours will directly influence incentive compensation models by correlating successful engagement data to successful sales outcomes. Food and Drug Administration’s Office of Prescription Drug Promotion ( OPDP ).”.

MedCity News
OCTOBER 9, 2023
The opening day of the HLTH 2023 event offered a surprising announcement from venture capital firm General Catalyst. ENGAGE at HLTH offered compelling discussions around retail health, prescription drug prices and investment trends. Weight Watchers CEO called for a change in how we talk about obesity.

MedCity News
MARCH 14, 2023
CMS Administrator Chiquita Brooks-LaSure discussed CMS’ key focus areas for 2023. This includes the end of the public health emergency, health equity and prescription drug costs. During an interview at the AHIP Medicare, Medicaid, Duals and Commercial Markets Forum held Tuesday in Washington, D.C.,

PM360
MAY 15, 2023
She combines business problem-solving with a tremendously human-centered approach to their desired outcomes, examining a brand’s specific ability to impact access and speed along the patient prescription journey.” Laura is not shy about sharing her cancer experience.

pharmaphorum
DECEMBER 22, 2022
As this collective body of experience continues to proliferate, 2023 will see additional FDA guidance. In 2023, expect to see AI innovations improve enrolment agility and, most importantly, the patient experience in a clinical trial. Beyond 2023 – the Sovereign Patient ID.

pharmaphorum
JANUARY 27, 2023
It’s wise advice from the “Sage of Omaha”; indeed, one highly respected financial newspaper began its 2023 predictions by conceding that it had been wrong on five major counts for 2022. Demand has also increased due to changes in prescribing protocols, leading to a higher underlying number of prescriptions for certain antibiotic medicines.

PharmExec
APRIL 14, 2023
Using prescription digital therapeutics can lead to better outcomes for patients, making them a win-win for pharma and patients.

Nixon Gwilt Law
FEBRUARY 27, 2023
Late Friday (February 24, 2023), the US Drug Enforcement Agency (DEA) released a proposed rule to make permanent some of the flexibilities allowed during the Public Health Emergency (PHE) for telemedicine companies whose providers prescribe controlled substances. Now let’s get to it.

MedCity News
MARCH 7, 2023
WeightWatchers will acquire Sequence, a platform that allows patients to have virtual appointments with a clinician, gives access to a dietitian and fitness coaching and provides prescription medications, including Ozempic and Wegovy.

PM360
DECEMBER 14, 2023
Survey any physician if they’ve been asked by a patient about a prescription weight loss medication and you’ll hear a resounding “yes.” The recent Sermo Barometer confirms that 89% of 300+ global practitioners have seen an increase in patients proactively requesting prescription weight loss medications.

Pharmaceutical Commerce
NOVEMBER 10, 2023
Blue Fin Group exec speaks on his upcoming feature.
Eversana Intouch
JANUARY 19, 2023
Success in 2023 will mean addressing each of those five issues meaningfully – understanding them fully and giving them real action, not just lip service. Pharma marketers in 2023 need to: Look for blind spots. Each year, we forecast several trends that will influence life-sciences marketing the most in the months to come ahead.

MedReps
JANUARY 30, 2023
Medical Sales Trends to Watch Out For in 2023 If you want to know what the future holds and create some forward-thinking sales goals, then check out these medical sales trends to watch out for in 2023. Along with an increase in elderly people, there’s also more healthcare spending taking place.

PM360
JULY 13, 2023
What if the industry were to incentivize persistence over new prescriptions? NRx data drives stock prices, determines ROI, and directs bonuses. What if instead of short-term gains, we prioritized the long game? Imagine if we collectively proclaimed that the real value is not only in starting, but in staying.

PM360
OCTOBER 13, 2023
It seems fewer doctors guide patients to intermittent fasting (62%) or anaerobic exercise (55%) than to prescription weight loss drugs and aerobic exercise. InCrowd also uncovered some other surprising doctor perceptions.

PM360
MAY 15, 2023
Karina joined OptimizeRx in 2021, where she has increased the digitalization of access to help move drug manufacturers away from manual processes.

PM360
APRIL 7, 2023
This is especially significant for manufacturers of high-cost specialty drugs, who will now face a recurring additional 20% charge on virtually every prescription fill. On the other side, drug manufacturers are facing the squeeze, with caps on price inflation and a mandated 20% discount in the catastrophic phase.

MedCity News
FEBRUARY 4, 2024
In 2023, prescription drug costs rose by 8.4%, a 31% increase from the prior year. By enforcing transparency, PBMs would need to release detailed information on prescription drug spending. 2024 stands at a critical juncture in the battle against soaring pharmacy costs. Transparency and reporting requirements.
Pharmacy Times
AUGUST 18, 2023
Based on results from a trial of repotrectinib in patients with ROS1-positive locally advanced or metastatic non-small cell lung cancer, the FDA accepted the New Drug Application and set a Prescription Drug User Fee Act goal date of November 27, 2023.

World of DTC Marketing
MARCH 10, 2022
The cost of Humira, which is injected via syringe, was more than $72,000 a year on prescription drug websites this week and is not expected to come down until at least 2023. The increase followed total Humira price hikes of 19 percent during 2017 and 2018. The executives at AbbVie make over $300 million.

European Pharmaceutical Review
OCTOBER 24, 2023
Contributing factors include greater demand for prescription drugs , advancements in the designing and printing of labels, high investments as well as pharmaceutical sales, plus high research and development (R&D) expenditures on pharmaceuticals and government initiatives. percent between the forecast period: 2023 and 2028.

Pharmaceutical Technology
JUNE 21, 2023
The company expects to launch LODOCO for prescription use in the second half of 2023. “We are dedicated to addressing heart disease, the leading cause of death, by ensuring all patients have access to LODOCO.”

Pharmacy Times
MAY 23, 2023
The FDA’s final decision is expected by a Prescription Drug User Fee Act goal date in August 2023.

Pharmacy Times
DECEMBER 21, 2022
The FDA granted Priority Review to both applications and set a Prescription Drug User Fee Act goal date for each application of April 21, 2023.
Pharmaceutical Technology
MAY 25, 2023
The US Food and Drug Administration has set 23 September 2023 as the prescription drug user fee action date for ATI-1501.

Pharmaceutical Technology
JUNE 12, 2023
Patients with erectile dysfunction (ED) will now be able to access a topical treatment without the need for a prescription after the US Food and Drug Administration (FDA) greenlit Futura Medical’s Eroxon. Futura says it plans to continue launches throughout 2023. ED affects approximately 18% of men in the US.

Clarivate
APRIL 18, 2024
Amazon’s big splash happened in February 2023 when it purchased the national primary-care practice One Medical for $3.9 RxPass offers more than 50 select prescriptions at no additional cost for $5 per month. billion, following an earlier failed healthcare venture with Berkshire Hathaway and JPMorgan Chase.
Pharmaceutical Technology
MAY 2, 2023
The regulatory body has also granted priority review to the application and set 28 August 2023 as a target date for the prescription drug user fee act (PDUFA). The European Medicines Agency (EMA) has also validated the Type II variation application for Reblozyl.

Pharma Leaders
MARCH 26, 2023
have introduced the Affordable Insulin Now Act of 2023, which would cap out-of-pocket insulin costs at $35 a month for both insured and uninsured individuals. Raphael Warnock (D-Ga.) and John Kennedy (R-La.) This bill is a follow-up to a bill Sen. The bill was signed into law in August 2022.

Pharmaceutical Technology
MAY 16, 2023
In January 2023, the US Food and Drug Administration (FDA) approved lecanemab under the accelerated approval pathway to treat AD. The regulator has also accepted Eisai’s supplemental biologics licence application (sBLA) for lecanemab and granted priority review, with 6 July 2023 as the prescription drug user fee act (PDUFA) action date.

Contrarian Sales Techniques
FEBRUARY 24, 2023
The company also offers online prescriptions and delivery services for medications. MyDoc - MyDoc is a telemedicine company that offers virtual consultation services with certified doctors, as well as online prescription services and medication delivery. The company also provides corporate healthcare solutions for businesses.
Pharmaceutical Technology
MAY 26, 2023
The FDA has now assigned a prescription drug user fee act action date of 23 November 2023 for the NDA.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content