Jacobio plans bid for China’s KRAS market with safety edge
pharmaphorum
MAY 1, 2024
China's Jacobio Pharma is preparing to file its KRAS inhibitor glecirasib for approval, saying it could offer a safety advantage over current drugs in the class
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

pharmaphorum
MAY 1, 2024
China's Jacobio Pharma is preparing to file its KRAS inhibitor glecirasib for approval, saying it could offer a safety advantage over current drugs in the class

European Pharmaceutical Review
MARCH 27, 2024
A report by Allied Market Research has predicted that the global pyrogen testing market will value $3.3 The market is predicted to grow at a CAGR of 8.2 A higher incidence of infectious diseases is a major contributing factor for the expansion of the pyrogen testing market. billion by 2032.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
MAY 8, 2024
After bringing slow-release schizophrenia drug Uzedy to market last year, Teva and France’s MedinCell are using the same playbook for a similar candidate. |

Fierce Pharma
JULY 12, 2023
Fighting for a place in the competitive atopic dermatitis market, Incyte hopes to reach younger patients with its JAK inhibitor cream Opzelura. Fighting for a place in the competitive atopic dermatitis market, Incyte hopes to reach young patients with its JAK inhibitor cream Opzelura.

World of DTC Marketing
JULY 21, 2021
SUMMARY: DTC marketing is not the reason why prescription drugs cost so much. The voices in Washington DC are once again calling for a moratorium on DTC marketing but lawmakers need to understand what DTC marketing actually does as upped to what people believe it does. DTC ads raise awareness around health conditions.

Pharma Marketing Network
MAY 3, 2023
Pharmaceutical marketing is a critical aspect of the industry, as it helps in spreading awareness about the benefits and potential side effects of different drugs. However, pharmaceutical marketing is also regulated by several agencies to ensure that patients are not exposed to false or misleading information. References: 1.
European Pharmaceutical Review
APRIL 17, 2024
A report by Market Research Future has predicted that the rapid sterility testing market will increase in value from $2.3 In the rapid sterility testing industry, the membrane filtration segment dominated the market , the author stated. Therefore, it is also contributing to growth of the rapid sterility testing market.
European Pharmaceutical Review
APRIL 11, 2024
Key drivers for the market include patent expirations of several biologic drugs and increasing demand for affordable biologic treatments, the author explained. Over the past few years, the biosimilar market has expanded “rapidly” due to these medicines offering a cost-effective alternative to the reference product.

Pharmaceutical Technology
NOVEMBER 1, 2022
FEATURED COMPANIES COMMENDED : • Innovation • Product Launch • Safety VIEW PROFILE COMMENDED : • Business Expansion • Innovation • Investments VIEW PROFILE COMMENDED: • Social VIEW PROFILE. It also includes projects and initiatives by companies to improve health and safety in other public environments other than their own workspaces.

Pharma Marketing Network
JULY 26, 2023
The Food and Drug Administration (FDA) plays a critical role in regulating the pharmaceutical industry and ensuring that medications and medical devices marketed to the public are safe, effective, and appropriately labeled.

LEVO Health
FEBRUARY 8, 2024
As healthcare professionals (HCPs) become more adept at engaging with digital marketing content , pharmaceutical marketers find it crucial to recalibrate their strategies. Therefore, pharma companies are investing heavily in digital marketing strategies to reach and engage with HCPs. and 8:00 p.m. on weekdays.

PharmExec
AUGUST 22, 2023
Amid Twitter's transformation into "X", social media expert Mikaela Walker weighs in on the rebrand's impact for pharma marketing, assesses ongoing brand safety risks, and highlights established social platforms and those gaining traction.

pharmaphorum
JULY 6, 2022
And how can regulators balance timely access with robust safety? Of the 23 that received conditional marketing authorization in Europe, 10 (43%) went on to receive regular approval, and just one (4%) was withdrawn. The ultimate goal, everyone agrees, has to be accelerating access while still maintaining the highest levels of safety.

European Pharmaceutical Review
FEBRUARY 8, 2024
A report from Prophecy Market Insights on rapid microbiology testing has predicted that the market is anticipated to surpass $12.7 Growth drivers of the global rapid microbiology testing market A rise in infectious diseases worldwide has increased demand for rapid and precise diagnostics. billion by 2034.

European Pharmaceutical Review
JANUARY 22, 2024
The global extractable and leachable (E&L) testing services market is projected to value $2.57 Crititically, they also enable a lower cross-contamination risk, enhancing patient safety. This this demand is a factor in the expected expansion of this market, the research highlighted. percent, the authors stated.

PharmExec
OCTOBER 17, 2022
October 18th 2022 9 am CT |10 am ET | 3 pm BST | 4 pm CET Oracle’s market-leading safety software solutions provide highly powerful technology and algorithms for data mining.

World of DTC Marketing
MARCH 16, 2021
IN BRIEF: As a marketer, most of us, like a Jungian archetype, are true Communicators. As a pharma marketer who has worked in many other therapeutic categories and recently on a Covid launch, I have become acutely aware of the interaction between national poles and the on-the-ground sentiment I have observed.

Pharmaceutical Technology
APRIL 29, 2024
PharmaLink has partnered with Recall Results for accurate and responsive drug recalls, withdrawals and market actions in the pharma industry.
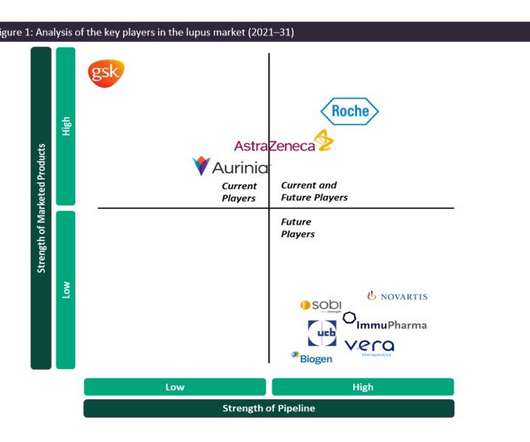
Pharmaceutical Technology
JANUARY 23, 2023
The management of SLE and LN consists largely of treatment options that are based on highly efficacious steroids or immune-suppressive therapy with an undesirable safety profile, which includes accrued organ damage, infections and cancer. in 2018 across the 7MM despite being an off-label therapy for lupus.
European Pharmaceutical Review
SEPTEMBER 22, 2023
According to a report by Research Nester, the global aseptic packaging market is projected to reach a revenue of $35 billion by 2033 and is estimated to reach a compound annual growth rate (CAGR) of 10 percent between 2023 and 2033. The post Aseptic packaging market to value $35bn by 2033 appeared first on European Pharmaceutical Review.
European Pharmaceutical Review
AUGUST 11, 2023
The European Commission has revoked the conditional marketing authorisation (MA) for Novartis’ sickle cell medicine crizanlizumab in the European Union (EU) and EEA. Conditional marketing authorisation is a pragmatic tool for the fast-track approval of a medicine that fulfils an unmet medical need.
European Pharmaceutical Review
AUGUST 21, 2023
According to a market report , the global RNA therapy clinical trials market is anticipated to reach $3.5 The data stated that expansion of the market in emerging countries is due to factors such as the rising success rate of RNA-based COVID-19 vaccines, regulatory approval, as well as demand for personalised medicines.

Pharma Marketing Network
JULY 19, 2023
The pharmaceutical industry is constantly evolving, and digital technology is playing an increasingly important role in marketing campaigns. In the past, pharmaceutical companies relied heavily on traditional marketing methods such as print ads, TV commercials, and direct mail.

eMediWrite
JULY 13, 2023
They play a crucial role in assuring the efficacy and safety of products given to patients. Since any inaccuracies might lead to the production of goods that might be ineffective or endanger human safety, it is understandable that regulatory oversight of QC Authority data is rigorous.

Clarity Engagement Solutions
NOVEMBER 9, 2020
The process of approval was simple: once the data showed the efficacy, safety and reliability to the regulatory agencies, the drug was approved. Thus, market access involved little engagement with a small set of stakeholders.In Thus, market access involved little engagement with a small set of stakeholders.In

Clarity Engagement Solutions
NOVEMBER 9, 2020
The process of approval was simple: once the data showed the efficacy, safety and reliability to the regulatory agencies, the drug was approved. Thus, market access involved little engagement with a small set of stakeholders.In Thus, market access involved little engagement with a small set of stakeholders.In – Channel.
Pharmaceutical Technology
APRIL 3, 2023
The European Commission (EC) has granted marketing authorisation for Sandoz’s biosimilar Hyrimoz (adalimumab) citrate-free high-concentration formulation (HCF). With eight marketed biosimilars, Sandoz is offering the broadest biosimilar portfolio and is the leading biosimilars company in Europe, with more than two decades of experience.

PM360
DECEMBER 20, 2023
Given China’s status as the world’s second most populous nation and an economic powerhouse, it represents both a monumental market opportunity and a dynamic arena of challenges for pharmaceutical products. From 2017 to 2022, the Chinese market witnessed a growth rate of 5.3%, compared to the average market growth of 6.6%

Pharma Marketing Network
OCTOBER 16, 2023
In this article, we’ll explore the power of social media in pharmaceutical marketing, and how to develop a successful strategy. It also provides an opportunity to share valuable information, such as product information, health advice, and safety warnings. This could include product information, health advice, and safety warnings.

PM360
JUNE 26, 2023
Traditionally, pharmaceutical companies have not prioritized emerging markets as part of their access strategy. A Complex and Challenging Environment When companies consider launching medicines in emerging markets, it might be tempting to suggest to simply lower the prices to reflect local affordability.
MedCity News
MARCH 31, 2024
Approval of Akebia Therapeutics’ Vafseo is based on additional post-marketing safety data from Japan, where the drug has been used since 2020. The FDA rejected the drug two years ago due to concerns about cardiovascular safety.

Pharma Marketing Network
AUGUST 9, 2023
The pharmaceutical industry operates in a highly regulated environment, where marketing and advertising practices are subject to rigorous scrutiny. In this blog post, we will explore best practices for pharmaceutical companies to navigate the complex landscape of regulatory challenges in marketing.

Pharma Marketing Network
JULY 19, 2023
Content Marketing as a Sales Driver: Unlocking Your Business Potential in the Pharmaceutical Industry Content marketing is a long-term strategy that can help businesses of all sizes generate leads, increase brand awareness, and drive sales.

Pharma Marketing Network
OCTOBER 7, 2023
Influencer marketing is a growing trend in the pharmaceutical industry, as companies look for new and innovative ways to reach their target audiences. As this trend increases, it is important to consider the ethical considerations and best practices for successful influencer marketing in the pharmaceutical industry.

pharmaphorum
NOVEMBER 22, 2022
GSK confirmed this morning that the disappointing readout from the DREAMM-3 trial of multiple myeloma therapy Blenrep reported earlier this month means that it will take the drug off the US market. Sales have been modest to date – just £36 million in the third quarter – despite it being the first drug targeting BCMA to reach the market.

Pharma Marketing Network
SEPTEMBER 15, 2021
Maximizing Patient Safety and Sustaining Long Term Quality Through Enhanced Device Surveillance while navigating the EU Regulatory Landscape. Discover how EU MDR changes requirements for Post Market Clinical Follow-up Studies (PMCF). Optimize Data and Databases for optimal Post Market and Post Clinic Surveillance and Quality.
European Pharmaceutical Review
DECEMBER 6, 2022
The European Medicines Agency (EMA) has accepted the marketing authorisation application (MAA) for tofersen, to treat superoxide dismutase 1 (SOD1) amyotrophic lateral sclerosis (ALS). Clinical data included in the marketing authorisation application. Sign up now !

Pharmaceutical Technology
OCTOBER 31, 2022
The results also demonstrated consistent efficacy against both RSV subtypes, RSV-A and RSV-B, as well as a favourable safety profile. If approved, GSK’s GSK-3844766A will be the first vaccine to reach the market for the prevention of RSV in older adults, gaining a significant first-mover advantage. in adults aged 70-79 years and 94.6%

LEVO Health
AUGUST 21, 2023
To leverage innovative B2B healthcare marketing strategies that resonate with this audience. This data underscores the need for a more tailored and strategic approach in B2B healthcare marketing. HCPs in the US might prefer content on safety and efficacy, while those in the UK lean towards real-world evidence. The challenge?

European Pharmaceutical Review
DECEMBER 5, 2022
GSK has announced that the European Medicines Agency (EMA) has validated the marketing authorisation application (MAA) for momelotinib, a new oral treatment for myelofibrosis, which, if approved, could be the only medicine available to address myelofibrosis, including anaemia, symptoms, and splenomegaly.

Pharmaceutical Technology
MAY 5, 2023
The European Medicines Agency’s (EMA) committee for medicinal products for human use (CHMP) has recommended full marketing authorisation (MA) for Gilead Sciences ’ Hepcludex (bulevirtide) to treat chronic hepatitis delta virus (HDV) and compensated liver disease in adult patients.
Pharmaceutical Technology
NOVEMBER 2, 2022
The European Medicines Agency (EMA) has granted full marketing authorisation (MA) for AstraZeneca ’s Covid-19 vaccine, Vaxzevria (ChAdOx1-S [Recombinant]). The vaccine initially received a conditional marketing authorisation (cMA) for use in the European Union (EU).

Pharma Marketing Network
JANUARY 6, 2021
Throughout the year 2020, pharma marketers were forced to explore new strategies in order to keep up with the many evolving trends. Pharma marketers have quickly learned how to adapt to these trends and are prepared to take on new opportunities in 2021. For example, people mistrust the efficiency and safety of the COVID-19 vaccine.
Pharmaceutical Technology
FEBRUARY 23, 2023
The market for UC is filled with various treatment options, and the pipeline is relatively strong. These novel agents are expected to strengthen the market by bringing more innovation to the space. The post New pipeline agents expected to differentiate the UC market appeared first on Pharmaceutical Technology.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content