Activ Surgical Secures CE Mark Approval for ActivSight™ Intelligent Light
Legacy MEDSearch
DECEMBER 6, 2022
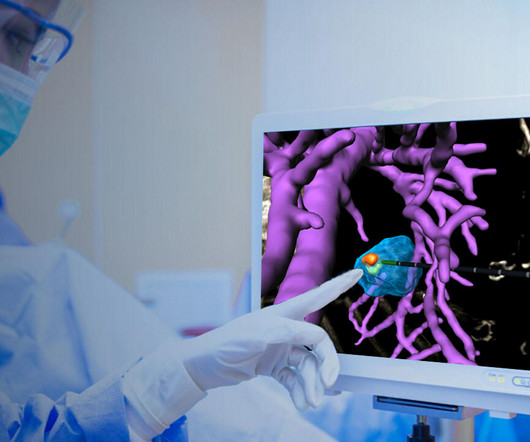
With ActivSight, surgeons can access critical intraoperative visual data as augmented reality overlays, helping to increase surgical outcomes and patient safety. “As We believe ActivSight will be a game-changer in the operating room by revolutionizing surgical vision and, ultimately, improving surgical care and patient safety.



























Let's personalize your content