The current pharma business model is unsustainable
World of DTC Marketing
SEPTEMBER 7, 2022
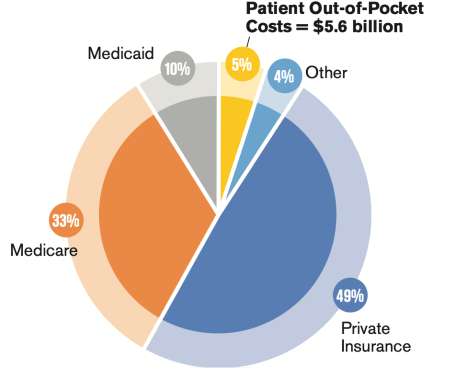
In pharma, growth depends on new products with hefty price tags when over 80% of voters want lower costs for their prescription drugs. Biogen’s failure should be a warning to other pharma companies. They relied so much on a new drug that they tried to market a product with bad science behind it. ” The U.S.




































Let's personalize your content