A physician’s perspective on health equity, diversity and inclusion
PharmaVoice
APRIL 24, 2023
Dr. Preeti Parikh sees parallels between promoting health equity and current DE&I challenges in the workplace.

PharmaVoice
APRIL 24, 2023
Dr. Preeti Parikh sees parallels between promoting health equity and current DE&I challenges in the workplace.

PM360
APRIL 25, 2023
How can countries be better prepared for the next pandemic? How do they need to change their public health infrastructure to ensure they don’t face similar challenges as posed by COVID-19? What can countries learn from each other to get these more modernized systems in place to improve their ability to exchange public health data across the globe? These questions were at the heart of many discussions during the 2023 Healthcare Information Management System Society (HIMSS) Global Health Conferen
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
APRIL 24, 2023
Apellis hits setback in ALS trial after pair of pegcetacoplan approvals fkansteiner Mon, 04/24/2023 - 10:56

MedCity News
APRIL 23, 2023
The healthcare sector has been notoriously slow to adopt new technologies in the past, but Chat-GPT has already begun to enter the field. Technology experts at the HIMSS conference in Chicago said that while the AI model is certainly exciting, the healthcare sector must establish an accountability framework for it’s going to address the risks of new technologies like ChatGPT moving forward.

Advertisement
Quotas need to be hit. Revenue goals need to be met. This reality makes shortening sales onboarding time a top priority. Organizations with a standard onboarding process boost employee retention by 58% and increase productivity by 50%. Unfortunately, many companies struggle with inefficient processes that lead to high turnover and missed revenue opportunities.

Pharmaceutical Technology
APRIL 26, 2023
AstraZeneca , Teva Pharmaceuticals , and Novartis , and other companies, are expanding their reach in the asthma space by capitalizing on digital technologies to enhance the patient experience. But experts say the complete integration of these devices and platforms into the healthcare system remains to be seen. Dr. Amy Chan, senior clinical research fellow at the School of Pharmacy, University of Auckland, says digital inhalers have existed for almost 30 years.
European Pharmaceutical Review
APRIL 25, 2023
The R21/Matrix-M malaria vaccine, developed by the University of Oxford , has been approved for use in Ghana (13 April 2023) and was authorised for use in Nigeria on 17 April. [Regulatory] approval of the [R21/Matrix-M ] malaria vaccine in Ghana was the first in the world” Approval of the malaria vaccine in Ghana was the first in the world. Granted full national licensure by the country’s Food and Drugs Authority (FDA Ghana), it is authorised for children aged 5 to 36 months.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.
MedCity News
APRIL 25, 2023
In just 2020, deaths from gun violence cost the U.S. healthcare system $290 million, or about $6,400 per patient, according to a report from the Commonwealth Fund. These costs are mostly covered by Medicaid and other government insurance programs.

PharmaTech
APRIL 28, 2023
Todd Andrews, global director of Applications and Business Development at CPC, discusses sterile aseptic connectors, flexibility in manufacturing, and more in an interview held at INTERPHEX 2023.
European Pharmaceutical Review
APRIL 27, 2023
Immunotherapy treatment blinatumomab will now become standard treatment worldwide for babies with acute lymphoblastic leukaemia (ALL). This decision is based on new results from an international trial published in the New England Journal of Medicine. Blinatumomab significantly improved survival, with the rate increasing from 66 percent to 93 percent, compared to individuals just given prior chemotherapy.

Fierce Pharma
APRIL 25, 2023
FDA awards Biogen's ALS drug Qalsody an accelerated approval, following its experts' feedback this time aliu Tue, 04/25/2023 - 14:16

MedCity News
APRIL 26, 2023
Kaiser Permanente announced that it expects to acquire Pennsylvania-based Geisinger Health. Kaiser’s move is part of a larger plan — Geisinger will be the first health system to join Risant Health, a new company Kaiser launched to operate nonprofit health systems under value-based care models. Kaiser said it expects to invest $5 billion in Risant and fold five or six health systems into the company over the next five years.
Pharmaceutical Technology
APRIL 28, 2023
The US Food and Drug Administration (FDA) has granted an Orphan Drug Designation to Editas Medicine’s gene therapy EDIT-301 in sickle cell disease, based on an April 27 announcement. The US agency previously granted the Orphan Drug Designation to EDIT-301 for its study in beta thalassemia, in May 2022. The company is studying EDT-301 in sickle cell disease in a Phase I/II RUBY study (NCT04853576), and is on track to provide a clinical update by mid-2023.

Clarivate
APRIL 27, 2023
Our latest Global Research Report from the Institute for Scientific Information (ISI) , “U.S. research trends: The impact of globalization and collaboration,” raises important questions about how well past investment has prepared the U.S. scientific enterprise to achieve its goals. Our findings suggest that while the U.S. remains a leading science and technology power, it must acknowledge its shrinking domestic research capacity and work collaboratively with resourceful competitors to maintain i

Fierce Pharma
APRIL 26, 2023
FDA approves first oral microbiome therapy Vowst. Will Nestlé be around to help Seres with the launch?

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

MedCity News
APRIL 27, 2023
While the digital health sector might feel a little cramped in some areas, there are wide open spaces in others — and investors are ready to funnel money into companies looking to innovate these lesser-discussed aspects of care delivery and healthcare technology. Some of these areas include Medicaid, generative AI and patient-to-patient support.

Pharmaceutical Technology
APRIL 27, 2023
The European Commission’s (EC) long-anticipated pharma reform plans in the European Union have finally been unveiled , indicating a focus on improving access to medicines across the bloc while cutting down on market exclusivity. Described as the largest reform in over 20 years, the proposed revision touches on multiple topics ranging from unequal access to innovative medicines across the EU to new environmental protections.

European Pharmaceutical Review
APRIL 24, 2023
Included in this issue of European Pharmaceutical Review : FOREWORD New quality requirements for tobacco products David Elder, David P Elder Consultancy QA/QC MASS SPECTROMETRY The benefits of mass spectrometry for expediting biologics to patients Ian Anderson, Mostafa Zarei and Qifeng Zhang, Lonza IN-DEPTH FOCUS: Bioprocessing/Bioproduction Realising the potential of AAV gene therapies Rajiv Vaidya, Andelyn Biosciences What gene therapy manufacturers can gain from collaboration Neil Almstead,

Fierce Pharma
APRIL 27, 2023
Quitting Lilly's obesity drug tirzepatide may be difficult for many patients, exec says kdunleavy Thu, 04/27/2023 - 15:08

Advertiser: ZoomInfo
Marketing technology is essential for B2B marketers to stay competitive in a rapidly changing digital landscape — and with 53% of marketers experiencing legacy technology issues and limitations, they’re researching innovations to expand and refine their technology stacks. To help practitioners keep up with the rapidly evolving martech landscape, this special report will discuss: How practitioners are integrating technologies and systems to encourage information-sharing between departments and pr

MedCity News
APRIL 25, 2023
MedCity News connected with Integra Connect’s COO, Cory Wiegert at ViVE 2023 to discuss how their new platform will go beyond the “one size fits all” approach to data curation.

Pharmaceutical Technology
APRIL 24, 2023
Traditional medicine refers to the knowledge, skills and practices that indigenous and different cultures use to maintain health, encompassing forms such as herbal medicine, Traditional Chinese Medicine (TCM) and acupuncture. While 80% of the world’s population practices some form of traditional medicine, according to the World Health Organization (WHO), the practice is regularly discredited due to a lack of scientific support and regulation as well as evidence of negative side effects with cert

European Pharmaceutical Review
APRIL 24, 2023
Included in this Guide to Testing: Exploring low endotoxin recovery in drug products Chiara Celli, Marco Mingolla and Fabrizio Lecce from the Institute of Biomedical Research Antoine Marxer, discuss low endotoxin recovery in drug products analysed using the monocyte activation test. Sustainability in BET from your endotoxin experts Associates of Cape Cod International, Inc. explores the future of sustainable LAL testing.

Fierce Pharma
APRIL 26, 2023
UPDATED: Gearing up for key approvals, GSK projects 'Shingrix-like' sales for its RSV vaccine fkansteiner Wed, 04/26/2023 - 09:52

Advertisement
Are your sellers unsure about using AI in their day-to-day workflows? Or are they eager to try but uncertain where to start? They might be asking: Which tasks are best suited for AI? How will using AI affect my relationship with my customers? With so many tools available, which ones are the most useful to me? To help answer these and other common questions about using AI during the sales cycle, we surveyed B2B sellers who were early adopters to get their insight and advice about how to use gen A

MedCity News
APRIL 28, 2023
Amazon recently announced that it is shuttering its Halo division, a line of wearable health and fitness devices. This marks the third time in about two years that the e-commerce giant has closed one its healthcare businesses — Amazon Care failed last year, and Haven failed in 2021.

PM360
APRIL 25, 2023
No one disputes how much the media landscape has evolved over the last two decades. What has been discussed, at times very passionately, is the role and reputation of media in today’s environment. As marketing and communications professionals, we wanted to take a deeper look into how media is perceived, how social media has changed the game, and how we can best adapt to this ever-evolving dynamic.

European Pharmaceutical Review
APRIL 28, 2023
NEW, FLEXIBLE, data-driven manufacturing models are emerging to address the pharmaceutical industry’s need for greater operational efficiency. While new approaches aim to solve the unique challenges of the sector, like adhering to strict compliance regulations, digitalisation has quickly developed into a pathway for achieving information technology (IT)/operational technology (OT) integration, edge-to-enterprise connectivity, and overall operational excellence.

Fierce Pharma
APRIL 27, 2023
Pascal Soriot deflects shareholder discord over AstraZeneca's legal battles amid 'very encouraging' start to the year fkansteiner Thu, 04/27/2023 - 09:39
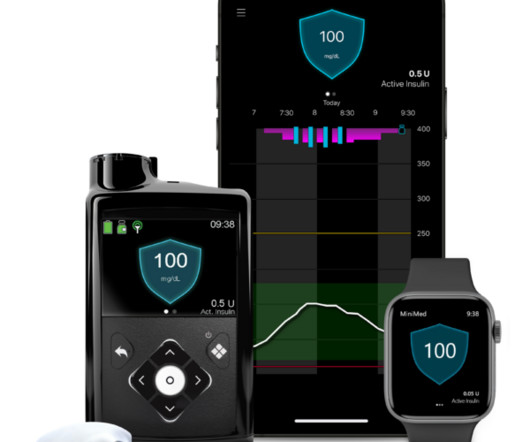
MedCity News
APRIL 25, 2023
Medtronic recently received FDA approval for its MiniMed 780G system, which is an insulin pump that automatically adjusts and corrects type 1 diabetes patients’ glucose levels every five minutes. The news comes two years after Medtronic submitted the system for FDA approval and three years after the company began selling the product in Europe.
Pharmaceutical Technology
APRIL 27, 2023
Seres Therapeutics and Nestlé Health Science have received approval from the US Food and Drug Administration (FDA) for Vowst (faecal microbiota spores, live-brpk) for preventing the recurrence of C difficile infection (CDI) in adults. Vowst is an orally administered microbiota-based therapeutic, previously known as SER-109, and is indicated for the prevention of recurrence of CDI in people aged 18 years and above following an antibacterial treatment for recurrent CDI (rCDI).

European Pharmaceutical Review
APRIL 28, 2023
A new global research consortium is being established to address the global impact of antimicrobial resistance (AMR). Centres for Antimicrobial Optimisation Network (CAMO-Net) , brings together research teams from the University of Liverpool and Imperial College London in the UK, the University of Cape Town in South Africa, the Infectious Diseases Institute in Uganda and the Faculty of Medicine at the University of São Paulo in Brazil. ”… this network… represents a major commitment t

Fierce Pharma
APRIL 28, 2023
EU plots major pharma reforms—but industry is already pushing back zbecker Fri, 04/28/2023 - 18:04

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.
Let's personalize your content