How to evolve artificial intelligence models alongside societal needs
MedCity News
OCTOBER 16, 2022
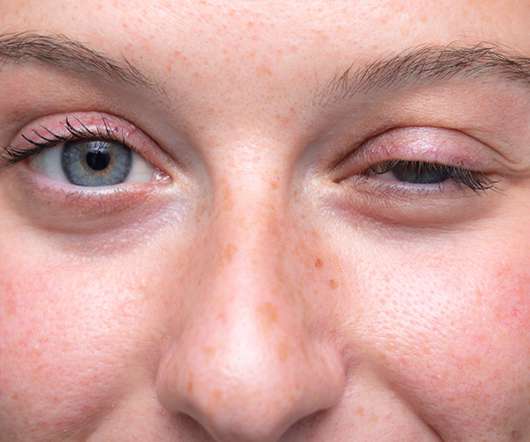
As society becomes more open to and reliant on these tools, it is the responsibility of technology companies, especially those tasked with assisting people’s mental health, to build and maintain artificial intelligence and machine learning models that adapt alongside societal needs.































Let's personalize your content