AZ, Merck's Lynparza wins FDA nod in prostate cancer subgroup following advisory committee vote
Fierce Pharma
JUNE 1, 2023
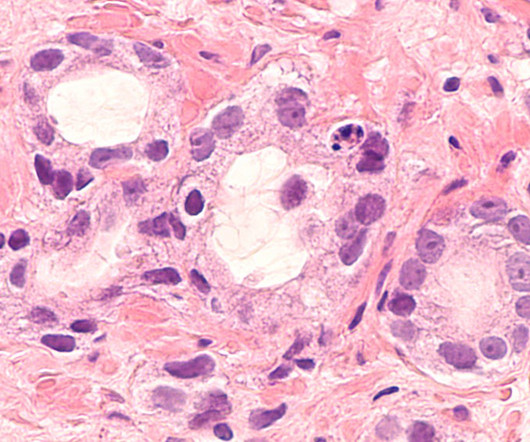
After a panel of independent experts endorsed a narrow approval for AstraZeneca and Merck’s Lynparza in metastatic castration-resistant prostate cancer (mCRPC), the FDA has followed suit. | The drug is now approved to treat BRCA-mutated metastatic castration-resistant prostate cancer in combination with Johnson & Johnson’s Zytiga and a corticosteroid.







































Let's personalize your content