Roche app relieves diabetes distress in clinical trial
pharmaphorum
OCTOBER 5, 2023
Roche app relieves diabetes distress in clinical trial Phil.Taylor Thu, 05/10/2023 - 07:39 Bookmark this
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 clinical diabetes
clinical diabetes 
pharmaphorum
OCTOBER 5, 2023
Roche app relieves diabetes distress in clinical trial Phil.Taylor Thu, 05/10/2023 - 07:39 Bookmark this
Fierce Pharma
FEBRUARY 6, 2024
In a midstage trial, Eli Lilly’s star diabetes and weight-loss drug tirzepatide showed promise in fatty liver disease, a difficult-to-treat condition that doesn’t yet have an FDA-approved therapy.<
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Fierce Pharma
JUNE 20, 2023
Three weeks after the FDA warned that some pharmacies are making unauthorized versio | Novo Nordisk filed five lawsuits in four states on Tuesday, charging health spas, clinics and pharmacies with false advertising, trademark infringement and unlawful sales of compounded versions of diabetes and weight loss drugs Ozempic and Wegovy.

MedCity News
FEBRUARY 11, 2024
Three big pharma companies joined the startup’s Series A financing, which will fund clinical trials in type 1 diabetes and myasthenia gravis. Cour Pharmaceuticals’ nanoparticles reprogram the immune system to stop inflammation behind autoimmune disease.

PharmExec
NOVEMBER 9, 2023
Healthy Food Rx provides home-delivered medical prescriptions of healthy food to help address diabetes.
Pharmacy Times
SEPTEMBER 8, 2022
Dual GIP/GLP-1 agonist therapy demonstrated superior glucose control and weight loss in patients with diabetes compared to selective GLP-1 receptor agonists in preclinical and clinical trials.

MedCity News
MARCH 24, 2024
A new report from the Peterson Health Technology Institute analyzed eight digital diabetes management tools. It found that the tools don’t provide meaningful clinical benefits and increase healthcare spending.

pharmaphorum
MARCH 22, 2024
First report from PHTI concludes that digital health technologies for diabetes management deliver minimal clinical benefit and raise healthcare costs

World of DTC Marketing
MARCH 29, 2022
Healthy eating is your recipe for managing diabetes. More than 37 million Americans have diabetes (about 1 in 10), and approximately 90-95% have type 2 diabetes. Type 2 diabetes most often develops in people over age 45, but more and more children, teens, and young adults are also developing it.

Fierce Pharma
JUNE 27, 2023
Diabetes patients in the United Kingdom may have to wait a bit longer to get their hands on Eli Lilly’s Mounjaro after the country’s drug value watchdog kiboshed its coverage recommendation.
European Pharmaceutical Review
DECEMBER 7, 2023
Results from a world-first human trial, published in the New England Journal of Medicine , has shown that a drug called baricitinib has promise as the first disease-modifying treatment of its kind for type 1 diabetes, that can be administered as a tablet.

European Pharmaceutical Review
MARCH 8, 2024
Study design The randomised, double-blind, parallel-group clinical trial compared injectable semaglutide 1.0mg with placebo as an adjunct to standard of care for prevention of progression of kidney impairment and risk of kidney and cardiovascular.

European Pharmaceutical Review
OCTOBER 18, 2023
TZIELD (teplizumab-mzwv) has demonstrated TZIELD’s potential to slow disease progression in newly diagnosed children and adolescents with Stage 3 type 1 diabetes. Data from the Phase III PROTECT clinical trial , presented at the 2023 Annual ISPAD Conference, showed that superior beta cell preservation was observed compared to placebo.

MedCity News
NOVEMBER 10, 2022
Sprinter Health is integrating its at-home clinical and diagnostic services into Firefly Health’s virtual primary care model. Firefly’s members will be able to receive common medical services from Sprinter clinicians in their homes — such as vital checks, blood draws, electrocardiograms, diabetic eye exams and diabetic foot screenings.
MedCity News
FEBRUARY 3, 2023
Despite challenging financial market conditions, clinical-stage Structure Therapeutics was able to upsize its IPO. Much of the new cash will support its lead program, an oral small molecule that could weigh in on the diabetes and obesity indications currently dominated by blockbuster injectable medicines.

European Pharmaceutical Review
DECEMBER 5, 2023
billion, enabling the Swiss pharma company to add a range of incretin treatments for obesity and diabetes to its portfolio. Carmot Therapeutics’ lead asset CT-388 is a Phase-II ready dual GLP-1/GIP agonist with best-in-class potential as a treatment for obesity in individuals with and without type 2 diabetes.

World of DTC Marketing
AUGUST 13, 2021
SUMMARY: Treating Americans with diabetes now costs more than the U.S. 85 percent of people with diabetes are overweight. While not all diabetes cases are linked to weight gain, overeating and eating too much is still the main cause of type 2 diabetes. Reuters this morning had a story on diabetes that scared me a lot.
World of DTC Marketing
JUNE 27, 2021
mg of semaglutide once weekly plus lifestyle intervention was associated with sustained, clinically relevant reduction in body weight” In the clinical study, participants were enrolled in an “intensive” behavioral study with 8 weeks low-calorie diet. We are an obese country, and diabetes is getting worse.

World of DTC Marketing
MAY 16, 2022
Food and Drug Administration approved Eli Lilly’s novel treatment for diabetes. The drug led to impressive blood sugar and body weight drops in clinical trials. The approval for tirzepatide—which Lilly will market under the brand name Mounjaro—isn’t for weight loss but Type 2 diabetes.
European Pharmaceutical Review
SEPTEMBER 8, 2023
Eli Lilly ’s Mounjaro (tirzepatide), has been recommended to treat poorly controlled type 2 diabetes in adults alongside diet and exercise. Compared with semaglutide , insulin therapy or a placebo, tirzepatide demonstrated that 81 percent to 97 percent of people achieved better glucose control in clinical trials.

European Pharmaceutical Review
OCTOBER 31, 2023
Insulin icodec, a once-weekly basal injection to treat type 1 diabetes, has the potential to be as effective in managing the condition as daily basal insulin treatments, according to results of the year-long ONWARDS 6 Phase IIIa clinical trial, recently published in the Lancet. Funding was provided by Novo Nordisk.

PharmExec
APRIL 7, 2023
Eli Lilly debuts its first Mounjaro TV commercial amid high demand for the diabetes injection, which also shows promise in weight loss applications and outperformed rivals Ozempic and Wegovy in clinical trials.
Pharmacy Times
DECEMBER 5, 2022
Teplizumab is a humanized IgG1? Fc-nonbinding anti-CD3 monoclonal antibody that blocks T cells, preventing them from attacking beta cells.

European Pharmaceutical Review
NOVEMBER 18, 2022
Tzield (teplizumab-mzwv), the first drug to help prolong the onset of stage 3 type 1 diabetes in adults and children over eight years old with stage 2 type 1 diabetes, has been approved by the US Food and Drug Administration (FDA). The drug binds to certain immune system cells and delays progression to stage 3 type 1 diabetes.
European Pharmaceutical Review
JUNE 27, 2023
Lonza and Vertex Pharmaceuticals Incorporated have agreed to collaborate to support the manufacture of Vertex’s Type 1 diabetes cell therapy portfolio. To support diabetes cell therapy production, construction of the US manufacturing facility is scheduled to begin later this year.

Pharmacy Times
JULY 20, 2023
Despite the surge in new diabetes cases, the treatment options in in children remain limited.
Pharmacy Times
OCTOBER 25, 2022
SGLT-2 inhibitors, GLP-1 receptor agonists, and a non-steroidal mineralocorticoid receptor are all showing promise in the prevention and management of diabetic kidney disease.

Pharmaceutical Technology
JANUARY 4, 2024
Tonabersat is under clinical development by Inflammx Therapeutics and currently in Phase II for Diabetic Macular Edema.
MedCity News
APRIL 28, 2023
Merck, Sanofi, and Eli Lilly joined the Series A financing of Therini Bio, a startup developing a drug that selectively targets fibrin as a way of reducing disease-driving inflammation in neurodegeneration and eye disorders such as diabetic macular edema. Therini is preparing to advance its lead program to clinical testing.
Pharmaceutical Technology
OCTOBER 7, 2022
Provention Bio and Sanofi US have signed a co-promotion agreement to launch the former’s lead investigational drug candidate, teplizumab for the delay in the onset of clinical type 1 diabetes (T1D). The US Food and Drug Administration (FDA) is presently reviewing teplizumab for the delay of clinical T1D in people who are at risk.
MedCity News
OCTOBER 17, 2022
Research shows that the company’s AI system is more accurate in detecting diabetic retinopathy than a clinical examination from a general ophthalmologist or a retina specialist.
Pharmaceutical Technology
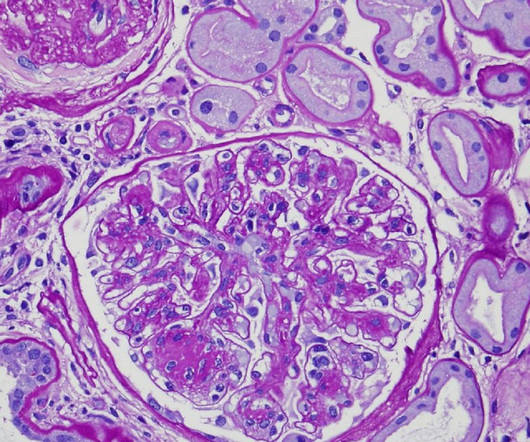
JUNE 7, 2023
The application was filed for the treatment of diabetic nephropathy/diabetic kidney disease. Glomerular diseases include focal segmental glomerulosclerosis, Alport syndrome and diabetic kidney disease. VAR 200 works by removing the extra lipids from the kidney. There are no therapeutic options available that address this issue.
European Pharmaceutical Review
DECEMBER 7, 2022
percent when used alongside other baseline treatments (diet, exercise, metformin and/or insulin), compared with placebo in a Phase III clinical trial of children and adolescents aged 10-17 years with type 2 diabetes. What is your opinion on Jardiance ® (empagliflozin) becoming another option for managing type 2 diabetes?

Pharmaceutical Technology
NOVEMBER 17, 2023
CH-0601 is under clinical development by Caelus Health and currently in Phase I for Type 1 Diabetes (Juvenile Diabetes).

European Pharmaceutical Review
AUGUST 14, 2023
Eli Lilly and Company has completed acquisition of two biopharmaceutical companies – Versanis Bio and Sigilon Therapeutics – and their lead assets focused on obesity and diabetes. Lilly announced the successful completion of its acquisition of clinical-stage Versanis Bio and its lead asset bimagrumab, an anti-obesity medicine.

European Pharmaceutical Review
DECEMBER 8, 2023
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has authorised diabetes medicine, Mounjaro (tirzepatide), for weight loss and weight management in adults aged 18 and over. The average percentage change in weight over the trial period was -16.0 percent for the 5mg dose, -21.4 percent for the 10mg dose, -22.5

Pharmaceutical Technology
APRIL 12, 2023
Clinical-stage biotechnology firm LyGenesis has formed a joint research collaboration with Imagine Pharma to develop new cell therapies for type 1 diabetes (T1D). In June 2022, the company discovered T1D AIPCs, a critical component to treat T1D through autologous cell transplantation.
European Pharmaceutical Review
JULY 26, 2023
The oral, once-daily, highly selective sodium-glucose cotransporter 2 (SGLT2) inhibitor is the first type 2 diabetes medicine to include cardiovascular death risk reduction data for patients with type 2 diabetes and cardiovascular disease in its label in several countries.

World of DTC Marketing
JUNE 29, 2022
Clinics offering debunked cancer treatments are still advertising on Facebook despite the company’s efforts to control medical misinformation. Recently there was a vibrant discussion about using supplements to control blood sugar in a diabetes group. Pharma, for the most part, has been dabbling in social media.

pharmaphorum
OCTOBER 4, 2022
Danish company Novo Nordisk have announced positive results from the ONWARDS 5 phase 3 trial, with once-weekly insulin icodec demonstrating superior reduction of HbA1 in people with type 2 diabetes, in combination with a dosing guide app. Diabetes is one of the most prevalent health conditions in the US, affecting over 11% of the population.
European Pharmaceutical Review
MARCH 14, 2024
Additionally, there have been notable clinical developments for the treatment of kidney disorders. This month, new data from Novo Nordisk has revealed that semaglutide could become the first GLP-1 treatment option for patients with type 2 diabetes and chronic kidney disease (CKD). Internet] Vera Therapeutics. cited 2024Mar].

LEVO Health
FEBRUARY 1, 2024
Patient Example In a diabetes medication campaign targeting patients based on their treatment history, lifestyle factors, and digital behavior, the campaign could segment audiences into subgroups such as newly diagnosed patients, long-term patients, and those at risk of diabetes.
European Pharmaceutical Review
MARCH 14, 2024
Additionally, there have been notable clinical developments for the treatment of kidney disorders. This month, new data from Novo Nordisk has revealed that semaglutide could become the first GLP-1 treatment option for patients with type 2 diabetes and chronic kidney disease (CKD). Internet] Vera Therapeutics. cited 2024Mar].

Pharmacy Times
JANUARY 3, 2024
Liraglutide, a glucagon-like peptide 1 receptor agonist, has shown a clinically and significant reduction in glycated hemoglobin for those with type 2 diabetes.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content