FDA accepts BLA for Pfizer’s haemophilia B gene therapy
Pharma Leaders
JUNE 27, 2023
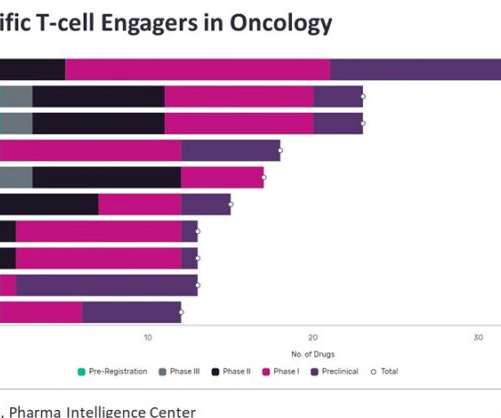
Based on the safety and efficacy findings from the Phase III BENEGENE-2 study, the company made these filings. Fidanacogene elaparvovec also demonstrated to be well-tolerated, with a safety profile in line with Phase I/II data. Pfizer obtained the licence for gene therapy from Spark Therapeutics in December 2014.





































Let's personalize your content