Developing a first-in-class small molecule drug for inflammatory disease
European Pharmaceutical Review
NOVEMBER 9, 2023
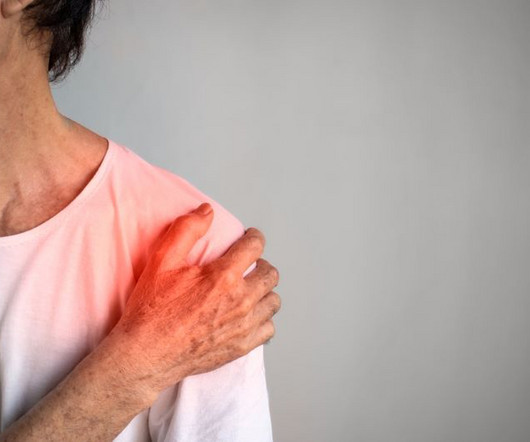
Following her presentation on age-related disease at BioFuture 2023, Executive Vice President of Drug Development for MyMD Pharmaceuticals ® , Jenna Brager, shares with EPR why the company’s next-generation tumour necrosis factor (TNF)-alpha inhibitor has potential in inflammatory and autoimmune disorders.

































Let's personalize your content