Pfizer warns Comirnaty sales will plummet by nearly two-thirds in 2023. And Paxlovid won't fare much better
Fierce Pharma
JANUARY 31, 2023
Pfizer warns Comirnaty sales will plummet by nearly two-thirds in 2023.

Fierce Pharma
JANUARY 31, 2023
Pfizer warns Comirnaty sales will plummet by nearly two-thirds in 2023.

European Pharmaceutical Review
JANUARY 31, 2023
A new strategy for 2023-2025 by the Medicines Patent Pool (MPP) aims to reduce access inequities, by improving the availability and affordability of health products in low- and middle-income countries (LMICs). The MPP is a United Nations-backed public health organisation working to increase medicine access and facilitate the development of life-saving medicines for LMICs.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
JANUARY 31, 2023
Amgen's Humira biosimilar Amjevita hits the market with two different list prices zbecker Tue, 01/31/2023 - 10:58
MedCity News
JANUARY 31, 2023
Orserdu, a drug from Menarini Group subsidiary Stemline Therapeutics, is now approved for treating breast cancers that carry the ESR1 mutation. The drug is the first approved oral therapy from a class of therapies called selective estrogen receptor degraders (SERDS).

Fierce Pharma
JANUARY 31, 2023
FDA extends review of argenx's subcutaneous efgartigimod, pushing decision date back into June ntaylor Tue, 01/31/2023 - 06:29
European Pharmaceutical Review
JANUARY 31, 2023
The European Commission (EC) has expanded the marketing authorisation for Dupixent® (dupilumab) in the European Union (EU) to treat eosinophilic esophagitis (EoE). It is the “… first and only targeted treatment option clinically proven to reduce both oesophageal inflammation and damage,” explained Dr Naimish Patel, Head of Global Development, Immunology and Inflammation at Sanofi.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

Legacy MEDSearch
JANUARY 31, 2023
Axonics, Inc., a global medical technology company that is developing and commercializing novel products for the treatment of bladder and bowel dysfunction, announced that the U.S. Food and Drug Administration has approved the company’s fourth-generation rechargeable sacral neuromodulation system. The newly approved Axonics R20 neurostimulator is labeled for a functional life in the body of at least 20 years and reduces how frequently a patient needs to recharge their implanted device to just on

Fierce Pharma
JANUARY 31, 2023
In mysterious FDA approval, Pfizer quietly wins Ibrance breast cancer expansion aliu Tue, 01/31/2023 - 10:37

LEVO Health
JANUARY 31, 2023
As the healthcare industry continues to evolve, digital marketing strategies, such as search engine optimization (SEO), have become increasingly crucial for healthcare organizations. Search engine optimization sits at the heart of any successful healthcare marketing strategy. Therefore, it is essential to become familiar with the role SEO strategy plays in healthcare marketing and healthcare SEO best practices that should be employed in 2023 to create a successful digital marketing plan.

Fierce Pharma
JANUARY 31, 2023
Waste not, want not: Aptar creates metal-free nasal spray pump to boost recyclability ntaylor Tue, 01/31/2023 - 10:56

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

MedCity News
JANUARY 31, 2023
Point solutions for areas like behavioral health, family planning and more have risen in popularity in recent years, experts said. But as employers deal with point solution fatigue, healthcare navigation companies are proving beneficial.

Fierce Pharma
JANUARY 31, 2023
The pharmacy channel is playing a critical role in marketing mteefey Tue, 01/31/2023 - 18:29

MedCity News
JANUARY 31, 2023
The final rule calls for stricter audits on Medicare Advantage plans. AHIP president and CEO Matt Eyles referred to the rule as “unlawful and fatally flawed.

Fierce Pharma
JANUARY 31, 2023
Seeking improved efficacy, Sensorion taps Eveon to deliver gene therapy to inner ear ntaylor Tue, 01/31/2023 - 10:23

MedCity News
JANUARY 31, 2023
Paradigm, a new startup co-incubated by ARCH Venture Partners and General Catalyst, recently launched with $203 million in Series A funding. The company is focused on improving equitable access to trials by creating a less fragmented and more efficient clinical research ecosystem.

Fierce Pharma
JANUARY 31, 2023
More modalities, more problems?
MedCity News
JANUARY 31, 2023
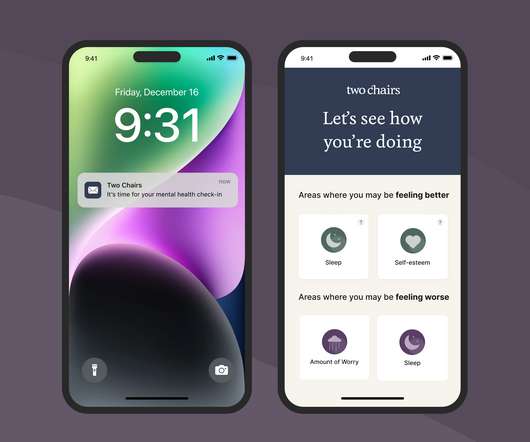
Less than 20% of behavioral healthcare providers collect data on how patients are progressing with their treatment. To buck this trend, Two Chairs is rolling out “mental health snapshots,” which are consistent summaries of patients’ well-being designed to offer real-time insight to both clinicians and clients.

MedReps
JANUARY 31, 2023
If getting into medical sales is your goal, then there’s a number of things you can do in order to make that dream come true, even if you’re still in college or have just graduated. How Students and New Grads Can Break into Medical Sales Continue reading to find out what college students and those who have recently earned their degree can do to successfully break into the medical sales field.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

MedCity News
JANUARY 31, 2023
Season one focusing on chronic disease management kicks off with a conversation with Stephanie Tilenius, CEO of VIda Health that aims to treat both the mind and the body.

Pharma Pathway
JANUARY 31, 2023
SunGlow Lifescience Pvt. Ltd-Walk-In Interviews for Production/ QC/ RA/ QMS/ ARD/ FRD/ Boiler Operator/ Maintenance/ Engineering/ Warehouse/ Regulatory Affairs/ QA On 5th Feb’ 2023 Job Description SunGlow Lifescience Pvt. Ltd was incorporated in January 2010 and was founded by a group of experienced industry professionals. Our Corporate office is located in Chennai, Indian.

MedCity News
JANUARY 31, 2023
Vector BioMed has developed a technology platform for designing and manufacturing the lentiviruses used to deliver cell and gene therapies. The startup is launching as the field continues to experience shortages of these viral vectors.

PharmaVoice
JANUARY 31, 2023
The biotech aims to complete rolling submissions with regulators in the U.K. and Australia in the first half of 2023.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharma Pathway
JANUARY 31, 2023
BDR Pharmaceuticals- Openings for Production/ QA/ QC/ CVC/ Packing/ Engineering- Apply Now Job Description BDR Pharmaceuticals was Established in 2002 into API manufacturing, We have a consolidated position in the domestic and global market for the API Business. In today’s dynamic global pharmaceutical market, we at BDR Pharmaceuticals have carved a niche for ourselves, with our formulations portfolio for specialty medication.

PharmaVoice
JANUARY 31, 2023
With the industry focused on cancer and COVID-19, Marc de Garidel made deals centered on cardio.

Pharma Pathway
JANUARY 31, 2023
Bliss GVS Pharma- Walk-Ins for Formulation Development On 4th Feb’ 2023 Job Description Walk-Ins for Research Associate/ Senior Research Associate/ Research Executive in Formulation Development @ Bliss GVS Pharma Departments: Formulation Development Designation: Research Associate/ Senior Research Associate/ Research Executive Experience: 01 to 08 years Job Location: Andheri, Mumbai Date : 4th Feb’ 2023 Time: 10:00 AM to 04:00 PM Venue: Bliss GVS Pharma Ltd. 302, Hyde Park, Saki Vih

PharmaTech
JANUARY 31, 2023
Be the Match BioTherapies’ and CIBMTR’s expanded service offering is intended to aid in the design, implementation, and oversight of cellular therapy clinical trials.

Advertisement
Clinical development organizations face a wide array of challenges when it comes to data, many of which can impact the operational effectiveness of their clinical trials. In this whitepaper, experts from Revvity Signals explore how solutions like TIBCO® Spotfire® enable better, more streamlined studies. The whitepaper also features a success story from Ambrx, a leading biopharmaceutical company, detailing how it has leveraged Spotfire to tackle data quality and collaboration challenges in clinic

Pharma Pathway
JANUARY 31, 2023
Rakshit Drugs Pvt. Ltd-Walk-In Interviews for QC/ Engineering/ R&D On 4th Feb’ 2023 Job Description Dear all Greetings from Rakshit drugs….! We are looking for below positions: Department: QC/ Engineering/ R&D Experience: 02 to 07 years Qualification: B.Sc/ M.Sc/ Diploma/ B.Tech (Mech.) No of Vacancies: 18 Walk-In Date: 4th Feb’ 2023 Time: 9:30 AM onwards Venue: Rakshit Drugs Pvt.

PharmaTimes
JANUARY 31, 2023
Phase 3 study demonstrated that efanesoctocog alfa met both primary and secondary endpoints

Pharma Pathway
JANUARY 31, 2023
Aurobindo Pharma-Walk-In Interviews for Quality Control On 3rd Feb’ 2023 Job Description Company Profile: Aurobindo Pharma Ltd’ (APL). APL is a growing India multinational pharmaceutical manufacturing firm with turnover of over US$2.8 Billion revenues for 2018-19, with presence in more than 34 countries fronted presence with products exported to 155 nations.

Copyright Clearance Center
JANUARY 31, 2023
The post A Copyright Reboot for Robots appeared first on Copyright Clearance Center.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
Let's personalize your content