Intravesical drug delivery system exhibits bladder cancer benefit
European Pharmaceutical Review
MAY 7, 2024
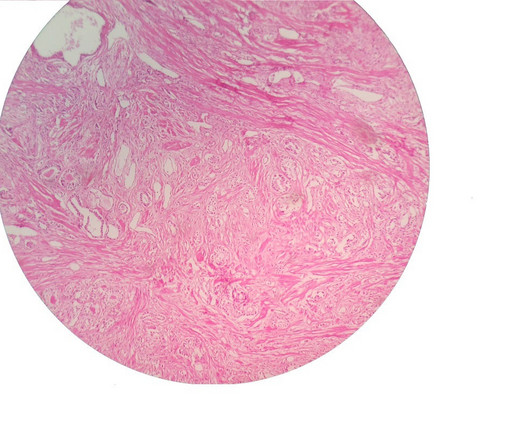
A novel intravesical targeted drug delivery system for patients with bladder cancer has demonstrated positive data in updated results from a Phase I trial. The treatment is being evaluated in patients with non–muscle-invasive bladder cancer (NMIBC) with select FGFR alterations.





























Let's personalize your content