Seeking full approval in Alzheimer's, Eisai dives into prominent Leqembi side effect
Fierce Pharma
MARCH 31, 2023
Seeking full approval in Alzheimer's, Eisai dives into prominent Leqembi side effect fkansteiner Fri, 03/31/2023 - 11:30
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
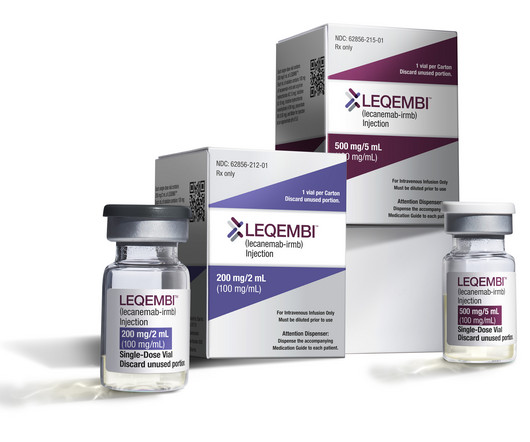
Fierce Pharma
MARCH 31, 2023
Seeking full approval in Alzheimer's, Eisai dives into prominent Leqembi side effect fkansteiner Fri, 03/31/2023 - 11:30

pharmaphorum
DECEMBER 4, 2023
Pfizer drops twice-daily obesity therapy due to side effects Phil.Taylor Mon, 04/12/2023 - 11:10 Bookmark this
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmatutor
JANUARY 23, 2023
Pharmacovigilance - Indian view of 2023 admin Tue, 01/24/2023 - 12:26 Pharmacovigilance is the process of monitoring and assessing the safety of medications after they have been approved and are on the market.

European Pharmaceutical Review
JANUARY 30, 2023
In its first meeting of 2023, the European Medicines Agency (EMA)’s Committee for Medicinal Products for Human Use (CHMP) recommended four new medicines for approval, providing positive opinions for two type 2 diabetes drugs. These side effects include cardiovascular conditions, blood clots, cancer and serious infections.

PM360
SEPTEMBER 21, 2023
Jeff Casberg, MS, RPh, Senior Vice President of Clinical Pharmacy at IPD Analytics, a managed care and pharmaceutical consultancy, highlighted a wide range of 2023 key events that will affect managed care professionals by the end of the year. This product is expected to have three generic launches in 2023.
European Pharmaceutical Review
JUNE 27, 2023
This data was presented at the 2023 Fetal Medicine Foundation (FMF) World Congress in Spain. Nipocalimab has been adapted through genetic approaches to optimise its clinical effect and minimise eventual side-effects.” In pregnancies requiring an IUT, all live-born infants required a simple transfusion.

PM360
DECEMBER 14, 2023
While side effects are anticipated with prescription weight loss medications (77% of physicians said their patients experienced side effects) an eye-opening 65% reported that their patients stopped taking the medication as a result of side effects.

pharmaphorum
JANUARY 6, 2023
In 2023, expect manufacturers to further shift their focus away from acquiring new patient prescriptions to retaining existing customers. Manufacturers can use 2023 as a time to examine these and other causes of patient drop-off in greater detail, and develop actionable solutions for each product in their portfolio. in 1979 to 2.8%in
European Pharmaceutical Review
APRIL 5, 2024
These interim data indicate early signs of potential clinical benefit with mRNA-3927, and importantly also demonstrate that mRNA-3927 has infrequent treatment-limiting side effects. Study data was presented at the 2023 American Society of Gene & Cell Therapy (ASGCT) Annual Meeting.
Pharmaceutical Technology
JUNE 5, 2023
Results unveiled at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting indicate that some patients with locally advanced rectal cancer can be treated with chemotherapy alone and may not need radiation therapy before surgery. In the FOLFOX group, 9.1% of patients received preoperative chemoradiotherapy and 1.4%

pharmaphorum
JULY 18, 2022
That is down to the company’s ADC technology platform, which means that the cytotoxic payload that circulates systemically after administration is lower than the already-marketed ADCs and should therefore limit side effects, according to the biotech.

PM360
OCTOBER 13, 2023
“They are effective but too expensive for the majority of patients, even with insurance coverage,” said one primary care physician (PCP). And while they’re excited about the drugs’ results and limited side effects now, many also noted concerns about the lack of safety data and long-term risks.

European Pharmaceutical Review
MARCH 14, 2024
HTL0048149 began first-in-human clinical trials in 2023, according to an announcement by Sosei in July 2023. Sosei Heptares will receive an upfront payment of €25 million.

European Pharmaceutical Review
APRIL 20, 2023
This is according to results from a preliminary study, of which the abstract will be presented at the American Academy of Neurology’s 75th Annual Meeting held between April 22-27 2023. AbbVie’s CGRP inhibitor Atogepant is a calcitonin gene-related peptide receptor antagonist, or CGRP inhibitor.

European Pharmaceutical Review
DECEMBER 22, 2023
This represents a 53 percent premium on Karuna’s closing stock price on 21 December 2023. It has demonstrated improvements in cognition and is not associated with common side effects of currently approved treatments like weight gain.

Clarify Health
OCTOBER 3, 2023
In drug research and personalized medicine, AI algorithms sift through intricate genetic information to recommend treatments tailored for individual patients, increasing the efficacy and reducing potential side effects.

European Pharmaceutical Review
NOVEMBER 3, 2023
The most common side effects of Litfulo are headache, diarrhoea, dizziness, acne, rash, inflammation of the hair follicles that may be itchy or painful and an increase in an enzyme called creatine phosphokinase, shown by blood test.

European Pharmaceutical Review
JUNE 1, 2023
These can have significant side-effects and can be ineffective for some people. NICE expects to publish the final guidance on rimegepant in June 2023. Current treatment options for preventing migraine include drugs used for other conditions such as beta-blockers, antidepressants and epilepsy medications.
pharmaphorum
SEPTEMBER 16, 2022
Minor side effects to long-term maintenance balance. Any clinical study seeks to achieve maximum results efficacy with minimal occurrence of side effects. However, side effects do happen. Regarding both common cold and headache, they are as well among the side effects reported for similar drugs.

Pharmaceutical Technology
NOVEMBER 14, 2022
Several other investigational therapies are expected to have material updates in 2023. In the five-patient report, mild cytokine release syndrome, a common side effect with CAR-Ts was observed, but the treatment was considered well tolerated.
European Pharmaceutical Review
APRIL 6, 2023
For adults with advanced prostate cancer it can also mean delaying chemotherapy and its associated side effects,” she added. NICE anticipates the final guidance on olaparib for breast cancer and prostate cancer will published in May 2023.

Pharmaceutical Technology
JUNE 5, 2023
“The treatments currently used to try to manage symptoms are associated with side effects and are often ineffective. In April 2023, the European Medicines Agency’s committee for medicinal products for human use recommended approval of Camzyos for the same indication.
European Pharmaceutical Review
JANUARY 11, 2024
The current medications (opioid and non-opioid) often force the patients to make an unfortunate choice between efficacy and debilitating side effects, leading to a cycling through multiple different therapies and suboptimal treatment response. Towards the end of 2023, one sensed an emerging interest from Big Pharma in neuroscience.

European Pharmaceutical Review
NOVEMBER 16, 2023
Side effects from treatment were similar to those associated with autologous stem cell transplants. The latest data from these ongoing trials for the CRISPR gene-editing therapy was presented at the European Hematology Association Congress in June 2023. No significant safety concerns were identified during the trials.

Pharma Leaders
MAY 18, 2023
The biopharmaceutical company expects XPHOZAH to be commercially available in Q4 2023 after an approval from the regulatory body. Diarrhea was the most common side effect with XPHOZAH. The regulatory agency has determined that the NDA is a class 2 review , which leads to a six-month review period from the date of resubmission.
Pharmaceutical Technology
APRIL 21, 2023
The company will make a $20m upfront payment, which includes $6m paid at closing and an additional $4m payment in the fiscal year 2023. US-based biopharmaceutical company Blue Water Vaccines has signed an asset purchase agreement with Veru to buy its Entadfi (finasteride and tadalafil) capsules business, for a total consideration of $100m.

pharmaphorum
OCTOBER 27, 2022
Santhera has completed a rolling application for its Duchenne muscular dystrophy (DMD) therapy vamorolone in the US, setting up a possible approval and launch in the latter half of 2023. “We look forward to working closely with US regulators to advance vamorolone towards approval,” he added.
European Pharmaceutical Review
OCTOBER 12, 2022
This is promising when side effects such as ARIAs are often detected in magnetic resonance imaging brain scans of patients who are given the antibodies. TauRx intends to submit its anti-tau therapy for regulatory approval in the US and Canada in 2023.

PM360
JULY 13, 2023
1 Patients may discontinue if they cannot afford their out-of-pocket costs even with insurance coverage due to side effects or lack of perceived need for the medication itself. Novartis’ analysis of claims data from Nov. 2020 to Oct. 2021 revealed that roughly one in four prescriptions were never filled.
Pharmaceutical Technology
MAY 26, 2023
It follows a March 2023 FDA Advisory Committee meeting where panelists voted 16-1 in favour of the drug’s approval. Paxlovid’s most common side effect is diarrhoea. This decision makes Paxlovid the first oral antiviral pill, and the fourth drug approved overall, to treat Covid-19 in adults.
European Pharmaceutical Review
JUNE 5, 2023
Nivolumab together with AVD chemotherapy is expected to become new standard for initial treatment for advanced stage disease in adolescents and adults, based on the trial data to be presented at 2023 American Society of Clinical Oncology (ASCO) Annual Meeting Plenary. Then 487 were part of the BV-AVD group.

Pharmaceutical Technology
JUNE 1, 2023
For context, in January 2023, the FDA accepted Intercept’s resubmission of its NDA for OCA as a Class 2 resubmission (which has a six-month review period in NASH) and set a Prescription Drug User Fee Act (PDUFA) date of 22 June 2023. risk reduction compared with placebo).
Pharmaceutical Technology
APRIL 14, 2023
Intercept’s New Drug Application (NDA) for obeticholic acid for use in patients with pre-cirrhotic liver fibrosis due to NASH, has a PDUFA date of June 22, 2023. Madrigal expects to file an NDA for the drug in H1 2023, based on a February 2023 company update. Caveats surrounding side effects for obeticholic acid remain.
European Pharmaceutical Review
NOVEMBER 9, 2023
Following her presentation on age-related disease at BioFuture 2023, Executive Vice President of Drug Development for MyMD Pharmaceuticals ® , Jenna Brager, shares with EPR why the company’s next-generation tumour necrosis factor (TNF)-alpha inhibitor has potential in inflammatory and autoimmune disorders.
European Pharmaceutical Review
OCTOBER 21, 2022
The most common side effects related to vaccination were injection site pain, malaise, fatigue, muscle aches and headaches. The international study will continue to examine the effects of two-dose HEPLISAV-B. The clinical trial is estimated to be completed in March 2023. Eight weeks after the second dose, 94.4

Pharmaceutical Technology
APRIL 24, 2023
While 80% of the world’s population practices some form of traditional medicine, according to the World Health Organization (WHO), the practice is regularly discredited due to a lack of scientific support and regulation as well as evidence of negative side effects with certain medicines.

European Pharmaceutical Review
JANUARY 25, 2024
They detail the medication’s intended use, proper administration, potential side effects and precautions. They are vital to ensuring the safe and effective use of medicines, making them an integral part of healthcare in Europe. References European Commission, (26 April 2023). Euractiv (6 October 2023).

Pharmaceutical Technology
JUNE 16, 2023
But while available adjunctive options work well, they come at the cost of safety, said Buntinx, and ANT01 could offer these benefits without additional side effects. ANeuroTech CEO Erik Buntinx ANeuroTech will enrol the first patient in its Phase IIIb trial of ANT01 in MDD in Q4 2023, said Buntinx.

pharmaphorum
AUGUST 31, 2022
The treatment also has a 10-minute onset of action that, according to Future, is “demonstrably faster” than the orally-dosed active comparator (tadalafil) used in the study, and also avoids the potential side effects of oral drugs for ED, which can take 30-60 minutes to take effect.
Pharmaceutical Technology
APRIL 24, 2023
The combination therapy also caused fewer side effects, with heart problems and hypertension as the most common ones, based on the final draft guidance. Brukinsa was approved by the US Food and Drug Administration (FDA) in CLL in January 2023. billion in 2029, while Calquence is expected to make $5.31

European Pharmaceutical Review
JULY 20, 2023
In June 2023, an array of leading voices in AAV development for cell and gene therapies articulated the current challenges and their optimism for the rapidly advancing sector at the Cell and Gene Therapy Summit event in London. More recently, the FDA granted approval of Roctavian in June 2023.

European Pharmaceutical Review
DECEMBER 14, 2023
As of October 2023, 62.93 They enable a range of observational study designs to be used where researchers study a period of exposure to a particular medicine and compare it with a control period of non-exposure in the same patient to establish any changes in the risk of adverse side effects. cited Dec2023].

pharmaphorum
OCTOBER 11, 2022
Leniolisib has been being given a speedy review for adults and adolescents aged 12 and over with APDS, with a decision due in the US towards the end of March and in the first half of 2023 in Europe. The filings are based on a 31-patient phase 2/3 trial which showed that the PI3K?

pharmaphorum
JANUARY 13, 2023
And as I look out the window at that snow, my travel-addled brain can’t help but see it as a metaphor for pharma in 2023: a crisp white, wide canvas laden with possibilities, but also one that’s going to require a lot of immediate, hard work from me if I want to be a responsible homeowner. Alright, throw me in metaphor jail. I deserve it.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content