Intercept's latest NASH bid in jeopardy after FDA questions drug's efficacy, safety
Fierce Pharma
MAY 17, 2023
Intercept's latest NASH bid in jeopardy after FDA questions drug's efficacy, safety aliu Wed, 05/17/2023 - 12:29
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Fierce Pharma
MAY 17, 2023
Intercept's latest NASH bid in jeopardy after FDA questions drug's efficacy, safety aliu Wed, 05/17/2023 - 12:29

pharmaphorum
NOVEMBER 28, 2023
FDA launches wide probe into CAR-T therapy safety Phil.Taylor Tue, 28/11/2023 - 18:27 Bookmark this
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PharmaTech
FEBRUARY 21, 2024
FDA's Drug Safety Priorities FY23 describes the center’s key safety programs and activities involved in promoting and protecting public health.

PharmaTech
SEPTEMBER 19, 2023
Maik Jornitz, Principal Consultant, BioProcess Resources LLC, discusses the definition of patient safety and how to implement new technologies into upgraded facilities.

European Pharmaceutical Review
MARCH 27, 2024
percent from 2023 to 2032, according to the research. For example, in 2023, Lonza Group launched several new solutions for endotoxin and pyrogen testing, the report highlighted. However, due to the importance of pyrogen testing for the safety of pharmaceutical products , the report stated that this will help to drive market growth.

European Pharmaceutical Review
JANUARY 29, 2024
N-Nitrosamines By far the biggest issue bedevilling industry during 2023 was the continuing N-nitrosamine contamination saga, which was covered in the third issue of EPR 2023. 4 The use of CPCA assessments allows for the assignment of scientifically justifiable higher acceptable intakes (AIs), without impacting patient safety.

European Pharmaceutical Review
MARCH 13, 2024
FDA inspections Identification of data integrity deviations Of the 70 Warning Letters issued by the US Food and Drug Administration (FDA) so far in 2024, three have identified data integrity issues at pharmaceutical manufacturing sites outside the US.
European Pharmaceutical Review
OCTOBER 18, 2023
The US Food and Drug Administration (FDA) has approved XPHOZAH ® (tenapanor), the first and only phosphate absorption inhibitor. XPHOZAH is expected to be available to eligible patients in the US in November 2023. The post First-in-class phosphate absorption inhibitor approved by FDA appeared first on European Pharmaceutical Review.

European Pharmaceutical Review
JUNE 26, 2023
US Food and Drug Administration (FDA) has published its first draft guidance presenting considerations to the pharmaceutical industry for designing clinical trials for psychedelic drugs. FDA’s draft guidance refers to psychedelics as “classic psychedelics”. These should be submitted latest 23 Aug 2023 to be considered.

Pharmaceutical Technology
APRIL 17, 2023
On 14 April 2023, experts from the US Food and Drug Administration’s (FDA) Advisory Committee (AdCom) voted largely in favour of the potential approval of Otsuka’ s and Lundbeck Pharmaceuticals’ Rexulti for the treatment of agitation associated with Alzheimer’s dementia (AAD). Rexulti is an atypical antipsychotic.
European Pharmaceutical Review
NOVEMBER 6, 2023
The Phase III data suggests that] IZERVAY is an effective and safe treatment option for patients with geographic atrophy” “The safety profile over two years was consistent with year 1, with no new safety signals identified. Astellas to accelerate ocular disease treatments with $5.9

European Pharmaceutical Review
JANUARY 20, 2023
The US Food and Drug Administration (FDA) has issued a complete response letter for the accelerated approval submission of donanemab for early Alzheimer’s, Eli Lilly and Company revealed. The safety profile of donanemab was initially reported from the TRAILBLAZER-ALZ trial in the New England Journal of Medicine.

Pharmatutor
FEBRUARY 2, 2023
FDA Inspections - Overview admin Thu, 02/02/2023 - 13:52 FDA Inspection is a regulatory process conducted by the United States Food and Drug Administration (FDA) to evaluate the compliance of food and drug establishments with FDA regulations and standards.

Pharmaceutical Technology
JUNE 19, 2023
UK-based pharmaceutical giant GSK has announced that the US Food and Drug Administration (FDA) has extended the review period of its new drug application (NDA) for the rare bone cancer drug momelotinib by three months. Novartis and Incyte Corp’s Jakafi was the first FDA-approved drug for the treatment of myelofibrosis in November 2011.
MedCity News
NOVEMBER 30, 2022
While an accelerated approval decision is expected in early 2023, the latest trial data are key because they represent the confirmatory study that could support an application for full FDA approval.
Pharmaceutical Technology
JUNE 9, 2023
Biopharmaceutical company Novaliq has received approval from the US Food and Drug Administration (FDA) for VEVYE (cyclosporine ophthalmic solution) 0.1% Novaliq medical science and regulatory affairs vice-president Sonja Krösser stated: “We are proud that the FDA approved VEVYE. to treat the signs and symptoms of dry eye disease.

European Pharmaceutical Review
JANUARY 10, 2023
Clarivate Plc has released its Drugs to Watch 2023 report — among 70 of the drugs highlighted, including potential blockbuster drugs, the majority were revealed to be personalised medicines. The report offers predictive analysis of drugs entering the market or launching key indications in 2023.

Pharmaceutical Technology
MARCH 2, 2023
The US Food and Drug Administration (FDA) has approved Reata Pharmaceuticals ’ oral, once-daily medication SKYCLARYS (omaveloxolone) to treat Friedreich’s ataxia patients. There are three more drug candidates with major trial readouts that are expected in 2023.
Pharmaceutical Technology
MAY 2, 2023
The US Food and Drug Administration (FDA) has accepted the supplemental biologics licence application submitted by Bristol Myers Squibb for Reblozyl (luspatercept-aamt) as a first-line treatment of anaemia in adults with lower-risk myelodysplastic syndromes (MDS).

pharmaphorum
DECEMBER 22, 2022
Prediction 1: the FDA will become more directive and collaborative on guidelines for DCTs. The risk of not having these discussions is to conduct expensive trials only to discover too late that they were measuring the wrong endpoint, in the wrong way, or with a tool or technology the FDA does not accept.
pharmaphorum
DECEMBER 19, 2022
The US Food and Drug Administration (FDA) has approved Swiss drugmaker Ferring Pharmaceuticals’ Adstiladrin (nadofaragene firedenovec-vncg) for the treatment of adult patients with high-risk Bacillus Calmette-Guérin (BCG)-unresponsive non-muscle-invasive bladder cancer (NMIBC) with carcinoma in situ (CIS), with or without papillary tumours.
Pharmaceutical Technology
JUNE 1, 2023
The US Food and Drug Administration (FDA) has granted a combination of AstraZeneca and MSD ’s Lynparza (olaparib), with standard therapies for treating BRCA-mutated (BRCAm) metastatic castration-resistant prostate cancer (mCRPC). Safety and tolerability were in line with that observed in prior trials and the known profiles of the medicines.
Pharmaceutical Technology
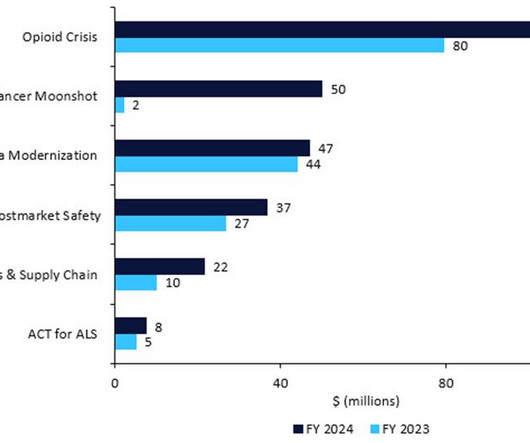
MARCH 30, 2023
The US FDA has revealed its detailed budget proposal for FY2024, which would require pharma companies to name their active pharmaceutical ingredient (API) suppliers, restart President Biden’s Cancer Moonshot, inject cash into amyotrophic lateral sclerosis (ALS) research, and enforce stricter rules around manufacturing, recalls, and shortages.
Pharmaceutical Technology
MAY 18, 2023
The US Food and Drug Administration (FDA) has accepted Ardelyx’s resubmission of a new drug application (NDA) for XPHOZAH (tenapanor) to control serum phosphate in adults with chronic kidney disease on dialysis who have had insufficient response or intolerance to a phosphate binder treatment.
pharmaphorum
NOVEMBER 23, 2022
The US regulator has started a priority, six-month review of Takeda’s dengue fever vaccine TAK-003 – tipped as a potential blockbuster product – making a decision likely in the first half of 2023. The post FDA starts speedy review of Takeda’s dengue vaccine appeared first on. billion in peak sales.
Pharmacy Times
MARCH 5, 2024
The next-generation SRI was previously granted an Orphan Drug Designation in November 2023 and its safety and efficacy are currently being evaluated in clinical trials.

PM360
SEPTEMBER 21, 2023
Jeff Casberg, MS, RPh, Senior Vice President of Clinical Pharmacy at IPD Analytics, a managed care and pharmaceutical consultancy, highlighted a wide range of 2023 key events that will affect managed care professionals by the end of the year. This product is expected to have three generic launches in 2023.

PM360
OCTOBER 13, 2023
Of the eight FDA-approved weight loss drugs on the market, these two are making waves for doctors who believe they represent “a new era for weight loss medications,” and “changing options and giving hope.” “They are effective but too expensive for the majority of patients, even with insurance coverage,” said one primary care physician (PCP).
Pharmaceutical Technology
MAY 31, 2023
The US Food and Drug Administration (FDA) has accepted Bristol Myers Squibb’s new drug application (NDA) for repotrectinib for priority review. The Phase I/II study assessed the pharmacokinetics, tolerability, safety and anti-tumour activity of repotrectinib in patients with advanced NSCLC.
European Pharmaceutical Review
FEBRUARY 21, 2023
SYFOVRE (pegcetacoplan injection) is the first and only treatment approved by the US Food and Drug Administration (FDA) for geographic atrophy (GA), a leading cause of blindness. The safety profile of SYFOVRE is well-demonstrated following ~12,000 injections. SYFOVRE is expected to be available by the beginning of March 2023.
Pharmaceutical Technology
MAY 26, 2023
After earning multimillion dollar revenues while being an authorised preferred treatment for Covid-19, the US Food and Drug Administration (FDA) has granted a full approval to Pfizer’s oral antiviral Paxlovid (nirmatrelvir + ritonavir). In response to the approval news, some pointed concerns about long-term safety.

Pharmaceutical Technology
JUNE 1, 2023
Additionally, when asked whether the FDA should consider a potential accelerated approval, panellists voted 15 to one against that regulatory pathway for the drug. Additionally, when asked whether the FDA should consider a potential accelerated approval, panellists voted 15 to one against that regulatory pathway for the drug.

Pharmaceutical Technology
MAY 11, 2023
The regulator accepted and gr anted priority review for the sNDA in January 2023. Brexpiprazole was found to be well-tolerated and its safety profile was consistent with its known safety profile in other indications. The change in agitation symptom frequency from baseline at week 12 was the primary endpoint of the two trials.
Pharmaceutical Technology
MAY 3, 2023
PharmaTher has submitted a fast track application for Ketarx (ketamine) to the US Food and Drug Administration (FDA) to treat levodopa-induced dyskinesia in Parkinson’s disease (LID-PD). PharmaTher is now assessing the design of the Phase III trial to align with the recommendations of the FDA.
Pharmaceutical Technology
MAY 25, 2023
Celltrion USA has received approval from the US Food and Drug Administration (FDA) for Humira (adalimumab) biosimilar, Yuflyma (adalimumab-aaty) , for multiple indications. Yuflyma represents the company’s fifth biosimilar and second anti-TNF biosimilar to receive US FDA approval.
Pharmaceutical Technology
APRIL 19, 2023
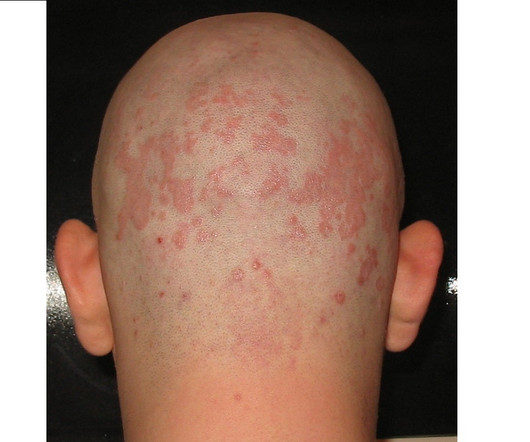
The US Food and Drug Administration (FDA) has accepted Arcutis Biotherapeutics ’ new drug application (NDA) for roflumilast foam 0.3% The regulator has set 16 December 2023 as a prescription drug user fee act (PDUFA) target action date for the decision on the application. in patients with seborrheic dermatitis.

Pharma Leaders
MAY 18, 2023
The US Food and Drug Administration (FDA) has accepted Ardelyx’s resubmitted New Drug Application (NDA) for XPHOZAH (tenapanor) to control serum phosphate in adult patients with chronic kidney disease on dialysis who have had an insufficient response or intolerance to a phosphate binder therapy.
Pharmaceutical Technology
APRIL 14, 2023
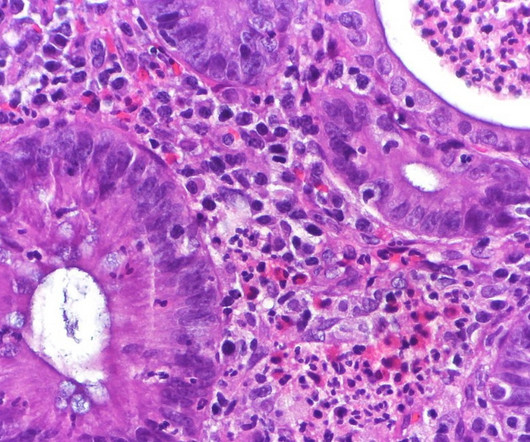
The US Food and Drug Administration (FDA) has rejected Eli Lilly’s biologic licence application (BLA) for the ulcerative colitis (UC) drug mirikizumab over manufacturing concerns. No concerns related to the clinical data package, safety or the medicine label. The regulator has issued a complete response letter.
PM360
APRIL 7, 2023
Med Device Department: Medicare Sets Precedent for Virtual Reality Therapeutic Payments In November 2021, the FDA approved a virtual reality (VR) therapy that provides an immersive experience for relieving chronic lower back pain. CMS factored in the efficacy of the device as well as its safety beyond the use of opioids.

Legacy MEDSearch
JANUARY 13, 2023
implantation of the HIT Reverse Hip Replacement System (Reverse HRS), under an FDA approved Investigational Device Exemption (IDE). The IDE Study is being conducted to determine the safety and effectiveness of the HIT Reverse HRS in Primary Total Hip Arthroplasty (THA). Press Release by: Hip Innovation Technology. Are you hiring?
Pharmaceutical Technology
NOVEMBER 1, 2022
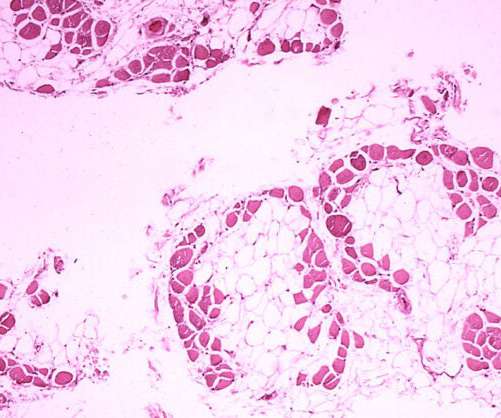
The US Food and Drug Administration (FDA) has granted Fast Track designation for Dyne Therapeutics’ DYNE-251 to treat Duchenne muscular dystrophy (DMD) mutations amenable to exon 51 skipping. In non-human primates, the product candidate showed a good safety profile.
Pharmaceutical Technology
APRIL 3, 2023
Tango Therapeutics has received clearance from the US Food and Drug Administration (FDA) for its investigational new drug (IND) application for TNG260 to treat STK11-mutant cancers. We look forward to initiating the Phase 1/2 clinical trial of TNG260 in the second half of 2023.”
pharmaphorum
SEPTEMBER 30, 2022
Patient organisations have been celebrating the FDA approval yesterday of Amylyx’ amyotrophic lateral sclerosis (ALS) therapy Relyvrio, after not one but two advisory committee meetings that arrived at different conclusions about the drug. The drug was also filed for approval in Europe in February, with a decision due in early 2023.

Pharmaceutical Technology
JUNE 9, 2023
Rocket Pharmaceuticals has received fast track and orphan drug designations for RP-A601 from the US Food and Drug Administration (FDA) to treat plakophilin-2-related arrhythmogenic cardiomyopathy (PKP2-ACM). In April 2023, the FDA approved the investigational new drug (IND) application for RP-A601 to begin a Phase I clinical trial.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content