Clinical Overview: Lecanemab Use, Coverage for Alzheimer Disease
Pharmacy Times
SEPTEMBER 26, 2023
In clinical trials, lecanemab showed a reduction of amyloid beta plaque in patients with Alzheimer disease.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 clinical alzheimer-disease
clinical alzheimer-disease 
Pharmacy Times
SEPTEMBER 26, 2023
In clinical trials, lecanemab showed a reduction of amyloid beta plaque in patients with Alzheimer disease.
Pharmacy Times
NOVEMBER 17, 2022
Patients with Alzheimer disease administered gantenerumab did experience slower clinical decline, however, it was not deemed statistically significant.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmacy Times
OCTOBER 11, 2023
Aducanumab, lecanemab, and donanemab are novel monoclonal antibodies designed to slow disease progression and hopefully have a positive impact in the field of Alzheimer disease management.
Pharmacy Times
OCTOBER 17, 2023
Lecanemab is a humanized immunoglobulin gamma 1 monoclonal antibody that targets amyloid plaques, which are thought to play a role in the development and progression of Alzheimer disease.
European Pharmaceutical Review
DECEMBER 8, 2023
Cerevel’s pipeline includes multiple clinical-stage and preclinical candidates with potential across several diseases including schizophrenia, Parkinson’s disease, and mood disorders. Gonzalez, chairman and chief executive officer of AbbVie. AbbVie also agreed a $10.1

MedCity News
DECEMBER 16, 2022
Unlike Alzheimer’s disease, which is characterized primarily by memory difficulties, but generally not major changes to personality or language, Frontotemporal dementia strikes at the core of what makes us who we are. Fortunately, there are several ongoing clinical trials addressing this disease.

Pharmaceutical Technology
APRIL 6, 2023
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has granted ADvantage Therapeutics’ immunotherapy AD04 an Innovation Passport for the treatment of Alzheimer’s disease. The difficulty in finding a treatment for Alzheimer’s disease has plagued the drug industry for decades.
European Pharmaceutical Review
OCTOBER 5, 2023
Longeveron’s Phase IIa trial of its stem-cell therapy for mild Alzheimer’s has revealed in top-line results, that certain doses enabled numerical slowing/prevention of disease worsening relative to placebo. Study safety data were consistent with an established safety profile.
European Pharmaceutical Review
JULY 17, 2023
Eli Lilly and Company’s TRAILBLAZER-ALZ 2 study has shown donanemab enabled nearly half of participants at earlier stage of Alzheimer’s to experience no clinical progression at one year, according to full results from the Phase III trial. Approximately half of participants met this threshold at 12 months.

PharmaTimes
SEPTEMBER 29, 2022
Results show reduction of clinical decline in global study of people with Alzheimer's Disease

Pharmacy Times
NOVEMBER 7, 2023
In May 2023, the FDA approved brexpiprazole as the first and only drug to receive approval for treatment of agitation associated with Alzheimer Disease dementia.
pharmaphorum
SEPTEMBER 29, 2022
Biogen and Eisai have resurrected hopes that amyloid-targeting drugs could have a benefit in Alzheimer’s disease with a claim that their new drug lecanemab showed a “highly statistically significant” reduction in clinical decline in a phase 3 trial.

World of DTC Marketing
JUNE 1, 2021
SUMMARY: The FDA is coming under intense pressure to approve Biogen’s Alzheimer’s drug, but Aaron S. Kesselheim, a professor of medicine at Harvard Medical School via the Washington Post , says “the worst thing for people with Alzheimer’s would be to put out a product that doesn’t work.”

European Pharmaceutical Review
JANUARY 11, 2023
Eisai has submitted a Marketing Authorisation Application (MAA) for lecanemab, for early Alzheimer’s (mild cognitive impairment due to Alzheimer’s (AD) and mild AD dementia) patients in Europe. Clarity AD study for Alzheimer’s. This application was submitted to the European Medicines Agency (EMA).

Pharma Marketing Network
MARCH 23, 2023
The Food and Drug Administration (FDA) has recently approved a new drug for the treatment of Alzheimer’s disease called Leqembi. This approval marks a major milestone in the fight against Alzheimer’s, which is the sixth leading cause of death in the United States and affects over 5 million people.

Pharmaceutical Technology
MAY 30, 2023
Lecanemab by Eisai Co Ltd, a monoclonal antibody therapy marketed for Alzheimer’s disease, is predicted to achieve the most significant commercial debut. which is the licensee of lebrikizumab, recently reported favourable outcomes for lebrikizumab through a novel secondary analysis of the Phase III clinical development programme.

Pharmacy Times
JANUARY 8, 2024
Those who were exposed to the clinically acquired stomach bacteria had a 24% increased risk of Alzheimer disease after a decade of onset.

Pharmaceutical Technology
FEBRUARY 19, 2024
Tonmya is under clinical development by Tonix Pharmaceuticals Holding and currently in Phase I for Alzheimer's Disease.
pharmaphorum
OCTOBER 7, 2022
TauRx Pharma’s effort to bring a tau-targeting drug for Alzheimer’s disease (AD) to market has been a marathon, with a number of setbacks over the years, but the company reckons it may finally be getting closer to the finishing line. As it turned out, that was a false expectation.

pharmaphorum
JANUARY 9, 2023
Eisai and Biogen’s Alzheimer’s therapy Leqembi will be priced at $26,500 per year – a little less than misfired predecessor Aduhelm – after getting conditional authorisation for the drug from the FDA on Friday. Aduhelm was launched at $56,000 per year, before a backlash prompted Biogen to slash it to $28,000.
Pharmaceutical Technology
MARCH 30, 2023
Biogen/Eisai’s newly approved drug, Leqembi (lecanemab), gained FDA approval in January 2023 for the treatment of Alzheimer’s disease. If approved in these regions, it would be established as the top-selling drug for Alzheimer’s disease in the coming years.

Pharmaceutical Technology
DECEMBER 25, 2023
HT-6184 is under clinical development by Halia Therapeutics and currently in Phase I for Alzheimer's Disease.

Pharmaceutical Technology
DECEMBER 20, 2023
PRI-002 is under clinical development by Priavoid and currently in Phase I for Alzheimer's Disease.
pharmaphorum
NOVEMBER 14, 2022
Roche’s Genentech division has reported disappointing top-line results from its highly-anticipated phase 3 trial of gantenerumab in early Alzheimer’s disease, in yet another setback for an amyloid-targeting drug. Study participants in GRADUATE 1 and 2 treated with gantenerumab showed a slowing of clinical decline of -0.31
European Pharmaceutical Review
NOVEMBER 8, 2022
The Gates Biomanufacturing Facility (GBF) announced the successful completion of the full cycle manufacturing of the recombinant Bulk Drug Substance (BDS) of the Alzheimer’s disease anti-Tau vaccine, AV-1980R, in collaboration with the Institute for Molecular Medicine (IMM), California. New therapeutics are needed.

Pharmaceutical Technology
FEBRUARY 9, 2023
Buntanetap tartrate is under clinical development by Annovis Bio and currently in Phase III for Alzheimer’s Disease. According to GlobalData, Phase III drugs for Alzheimer’s Disease have a 9% phase transition success rate (PTSR) indication benchmark for progressing into Filing rejected/Withdrawn.
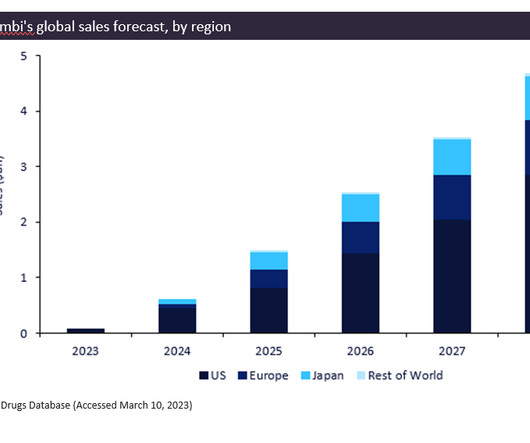
Pharmaceutical Technology
MARCH 30, 2023
Biogen/Eisai’s newly approved drug, Leqembi (lecanemab), gained FDA approval in January 2023 for the treatment of Alzheimer’s disease. If approved in these regions, it would be established as the top-selling drug for Alzheimer’s disease in the coming years.
Pharmacy Times
NOVEMBER 3, 2022
Eisai plans to file for traditional approval in the United States by March 31, 2023.

pharmaphorum
JANUARY 17, 2023
The deal extends Cambridge Cognition’s technology in the area of the use of voice-based assessments that can be used to detect and monitor health conditions, and according to the company gives it a platform “that can deliver most of the voice tasks used today by psychologists and neurologists to assess disease.”

Pharmaceutical Technology
NOVEMBER 5, 2023
AL-001 is under clinical development by Alzamend Neuro and currently in Phase II for Alzheimer's Disease.
European Pharmaceutical Review
MARCH 6, 2023
With nearly 10 million new cases of dementia of every year 1 , the increasing prevalence of Alzheimer’s disease (AD), the most common form of the condition, has prompted researchers and the pharmaceutical industry to seek novel, effective solutions. January 2023 marked a pivotal turning point for treating Alzheimer’s.

Pharmaceutical Technology
DECEMBER 15, 2022
Under the deal, the parties intend to develop treatments for diseases including Alzheimer’s and Parkinson’s. Louis, Eisai is working to fulfil our human health care mission and provide potential new and targeted disease-modifying therapies with the ultimate goal of achieving a world free of neurodegenerative disease.”.
pharmaphorum
JULY 11, 2022
AbbVie is pulling out of its collaboration with Alector on the development of Alzheimer’s disease candidate AL003, which aims to tackle the neurodegenerative disease by restoring reduced immune system activity in the brain. The post AbbVie cuts Alzheimer’s alliance with Alector in half appeared first on.
pharmaphorum
DECEMBER 22, 2022
One of the most highly-anticipated developments in pharma next year will be the FDA’s verdict on Eisai’s accelerated application for lecanemab in Alzheimer’s disease, due early in January. The post ICER says lecanemab data in Alzheimer’s “inconclusive” appeared first on.

pharmaphorum
AUGUST 2, 2022
In May, another amyloid targeting drug for Alzheimer’s disease – AC Immune and Roche’s crenezumab – missed the mark in another clinical trial and looked destined for the scrap heap like so many drugs in the class before it. So where is the sliver lining?

European Pharmaceutical Review
FEBRUARY 2, 2024
The company stated that it will discontinue the development and commercialisation of ADUHELM 100 mg/mL intravenous injection and will terminate the corresponding Phase III ENVISION clinical study. ADUHELM received accelerated approval from the US FDA in June 2021, Biogen confirmed.

pharmaphorum
NOVEMBER 14, 2022
Investors in TauRx Pharma have exercised warrants worth around $119 million, adding to the biotech’s financial resources as it prepares regulatory filings for tau-targeting Alzheimer’s drug hydromethylthionine mesylate (HMTM) on the back of its recently-reported LUCIDITY trial.
pharmaphorum
JANUARY 5, 2023
With the FDA due to deliver a decision of Eisai and Biogen’s new Alzheimer’s disease candidate lecanemab tomorrow, the safety of the drug has been thrust into the spotlight by a case report detailing a fatality in one of their clinical trials.

Pharmaceutical Technology
JUNE 2, 2023
The drug candidate has completed a Phase IIa and Phase IIb/III clinical trial for Alzheimer’s disease, a Phase II and a Phase III trial in Rett syndrome adult patients, and a Phase II proof-of-concept trial in Parkinson’s disease.

Pharmaceutical Technology
FEBRUARY 9, 2023
Buntanetap tartrate is under clinical development by Annovis Bio and currently in Phase III for Parkinson’s Disease. According to GlobalData, Phase III drugs for Parkinson’s Disease have a 46% phase transition success rate (PTSR) indication benchmark for progressing into Filing rejected/Withdrawn.
pharmaphorum
DECEMBER 30, 2022
The FDA’s interactions with Biogen in the build-up to last year’s controversial approval of Alzheimer’s disease therapy Aduhelm have been described as “inappropriate” and “atypical” in a congressional report on the review.

pharmaphorum
OCTOBER 6, 2022
A bipartisan bill has been introduced in the US Congress that will try to block Centers for Medicare and Medicaid Services (CMS) restrictions on Biogen’s Alzheimer’s therapy Aduhelm being extended to newer drugs for the disease.

pharmaphorum
JULY 6, 2022
The FDA has started an accelerated review of Eisai and Biogen’s a new amyloid-targeting antibody lecanemab for Alzheimer’s disease, with a 6 January deadline that keep them ahead of closest rival Eli Lilly. ” The post Eisai/Biogen could get FDA verdict on new Alzheimer’s drug by 6 Jan appeared first on. .
European Pharmaceutical Review
SEPTEMBER 7, 2023
In 2018, we found evidence of senescent cells in human Alzheimer’s,” stated Miranda Orr, PhD, Associate Professor of gerontology and geriatric medicine at Wake Forest University School of Medicine in the US. The post Alzheimer’s senescent cell combination therapy presents promise appeared first on European Pharmaceutical Review.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content