Biopharma industry lobbies for tax breaks related to US-based manufacturing
Fierce Pharma
MARCH 21, 2023
Biopharma industry lobbies for tax breaks related to US-based manufacturing kdunleavy Tue, 03/21/2023 - 11:14
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Fierce Pharma
MARCH 21, 2023
Biopharma industry lobbies for tax breaks related to US-based manufacturing kdunleavy Tue, 03/21/2023 - 11:14

Fierce Pharma
SEPTEMBER 11, 2023
The adoption of technology and data by biopharma is driving a digital transformation in manufacturing, accelerating the process of bringing molecules from benchside to bedside. I am the Strategic Business Unit Head and Global Head of Mergers and Acquisitions for Life Sciences at Cognizant. |
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
MAY 1, 2024
biopharma companies rely on Chinese contractors to provide lab research and drug manufacturing services. It’s no secret that U.S. Facing a push for China-U.S. decoupling from Washington, the industry trade group BIO now wants to quantify the U.S.’s s dependence on Chinese CDMOs through a survey.

European Pharmaceutical Review
NOVEMBER 16, 2022
As the emphasis of new drug development gravitates towards new, ground-breaking therapies and vaccines, the requirements of manufacturing sites, equipment and processes need to be appropriate for this new environment. That is because of the relative expense of emerging therapies and the pressure on pricing for continuing product lines.

European Pharmaceutical Review
JUNE 7, 2023
The global biopharma industry’s resilience has decreased since 2021, a report by Cytiva has found. Using feedback from 1250 pharma and biopharma executives in 22 countries, the research sought to find out how has the biopharma industry has evolved over the past two years and what areas the industry should seek to improve.

Fierce Pharma
MARCH 15, 2024
Over the years, Singapore has attracted a suite of biopharma majors, and Novartis is no exception. The project will introduce digitalization and automation at the site in a bid to boost manufacturing productivity and to help upskill the company’s workforce.

Fierce Pharma
JANUARY 4, 2024
Even after the heady highs of the COVID-19 era, the contract development and manufacturing organization (CDMO) realm remains one of biopharma's buzziest fields. Even after the heady highs of the COVID era, the CDMO realm remains one of biopharma's buzziest fields.

PharmaTech
JANUARY 9, 2024
Bradley Grobler, Vice President of Sales, Pharma & Biopharma Europe, describes how Actylis meets global demands for (bio)pharmaceutical development and manufacturing programs during CPhI 2023.

PM360
FEBRUARY 1, 2024
Embarking on the journey from product development to market success in the biopharma industry is no small feat. According to a McKinsey report , a startling 40% of biopharma products fail to meet their sales forecasts within the initial two years, prompting a critical reassessment of strategies for product launches.

European Pharmaceutical Review
JULY 7, 2023
New recommendations to boost the competitiveness in UK biopharma and Medtech sectors have been published by Imperial College London. Technological solutions and policy recommendations for government have been identified as a way to help make sectors such as biopharma and Medtech more innovative and increase their value added.

European Pharmaceutical Review
DECEMBER 20, 2023
1 The changes form part of the 2020 Pharmaceutical Strategy for Europe will impact pharma and biopharma companies operating in Europe. The first is removal of the RDP extension for pan-EU launch within two years, instead requiring manufacturers to submit to local P&MA processes upon request by the member state.

Fierce Pharma
JUNE 13, 2023
By: William Blake, Chief Technology Officer, Human-Based R&D | Danaher Life Sciences companies are ushering in a new era of engineering biology, partnering with biopharma leaders to deploy automation, AI and other engineering practices from early research through manufacturing.

Pharmaceutical Technology
SEPTEMBER 8, 2022
With our proven track record in cancer immunotherapy, we are well-positioned to leverage our deep discovery, development, clinical, and manufacturing capabilities and worldwide reach to potentially bring innovative products from this programme to patients as fast as possible.”.

Fierce Pharma
MAY 25, 2023
As drugmakers advance a wave of new cell and gene therapies, contract manufacturers have captured a larger share of the biopharma spotlight in recent years as they push to meet ever-increasing dema | As drugmakers advance a wave of new cell and gene therapies, contract manufacturers have captured a larger share of the biopharma spotlight in recent (..)

Pharmaceutical Technology
JULY 29, 2022
As the full consequences of the COVID-19 virus started to emerge, and many industries found themselves having to either suspend operations or move to remote working, the biopharma sector found itself on the front line facing unprecedented challenges. Manufacturing agility: Index score 6.5/10. R&D ecosystem - Index score 6.54/10.

Fierce Pharma
JULY 12, 2023
A new triumvirate of biopharma bigwigs is in line to reign over one of the industry’s top trade groups. | The International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) has unveiled the upcoming lineup for its CEO leadership team over the next two years.

MedCity News
NOVEMBER 19, 2023
The German manufacturing site is the latest capital infrastructure move for Eli Lilly, which has already made $11 billion in investments in its global manufacturing capabilities in the past three years. The new site will help Lilly meet strong demand for its diabetes and obesity drugs.

European Pharmaceutical Review
MAY 2, 2024
As a result, the market is expected to reach $719 billion by 2030, 1 Sigma Mostafa, PhD, Chief Scientific Officer at KBI Biopharma shared with EPR. These trends have an impact on manufacturing capacity. There are a lot of conjugated products in the clinical pipeline and novel conjugation partners and chemistries are being tried.

PharmaTech
APRIL 30, 2023
Technical Projects, Capacity Expansion Pharma Production shares details of Baxter BioPharma Solution’s approximately $100 million expansion of its sterile fill/finish manufacturing facility located in Halle/Westfalen, Germany. In this podcast, Kayleigh Hearse, Sr Mgr.
European Pharmaceutical Review
NOVEMBER 30, 2023
o find out about the current trends in cell and gene therapy manufacturing, EPR spoke to Cell and Gene Therapy Catapult (CGT Catapult)’s Chief Manufacturing Officer, Stephen Ward, and Chief Business Officer, Jeanette Evans. Licensing of the manufacturing suites is expected in early 2024. So now we have a really nice blend.”

PharmaTech
APRIL 30, 2023
Technical Projects, Capacity Expansion Pharma Production shares details of Baxter BioPharma Solution’s approximately $100 million expansion of its sterile fill/finish manufacturing facility located in Halle/Westfalen, Germany. In this podcast, Kayleigh Hearse, Sr Mgr.
Pharmaceutical Technology
DECEMBER 12, 2022
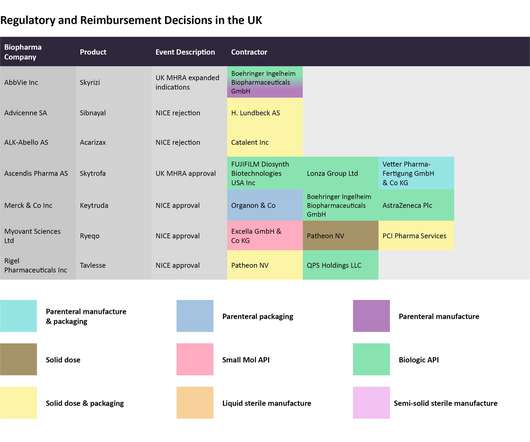
In this last 2022 edition of the series, which started in June , Pharmaceutical Technology is tracking major trial announcements and decisions by regulators and reimbursement agencies that have occurred since mid-October, as well as their potential impact on manufacturing plans. Iomab-B met the durable complete remission endpoint.

European Pharmaceutical Review
FEBRUARY 21, 2023
Crucially, January 2023 marked a key moment: the Medicines and Healthcare products Regulatory Agency (MHRA) granted Celadon Pharmaceuticals the first good manufacturing practice (GMP) registration since legalisation in 2018 for high Δ9-tetrahydrocannabinol (THC) cannabis active pharmaceutical ingredient (API).

European Pharmaceutical Review
JANUARY 12, 2023
Out of 19 companies honoured with the 2022 Terra Carta Seal, the three pharma companies awarded were: GlaxoSmithKline PLC. Minimising the use of disposables in the manufacturing process through waste management and recycling. ” Biopharma successful in achieving climate-related goals, report finds.
MedCity News
JANUARY 18, 2023
Cell therapy offers a treatment option for some advanced blood cancers, but the manufacturing process is lengthy and complex. The biopharmaceutical industry is working on various technologies that could make manufacturing faster and more efficient, or perhaps supplant the current process altogether.

pharmaphorum
DECEMBER 28, 2022
M&A is an integral part of the lifecycle of pharma companies and a key strategy to future-proof larger players, driving R&D activities and innovation for a competitive product pipeline. Since the first wave of consolidation started in 1988, there has been a significant concentration of the pharma industry.

European Pharmaceutical Review
SEPTEMBER 14, 2023
This includes its Macclesfield campus, the UK’s largest pharma manufacturing site. The pharma company’s sites in Macclesfield, Cambridge, Luton and Speke will be supplied by energy from the biomethane facility. Supply of the renewable gas is planned through a new 15-year partnership with Future Biogas.

Pharmaceutical Technology
NOVEMBER 30, 2022
However, efficiently manufacturing the drug represents another barrier to cross before realizing the full revenue potential then successfully. Each month, Pharmaceutical Technology takes a look at recent decisions taken by regulatory and reimbursement agencies and identifies the key manufacturing players that can be impacted by them.
Pharmaceutical Technology
MAY 9, 2023
Baxter International has signed a definitive agreement to sell its BioPharma Solutions (BPS) business to private equity investor Advent International and growth investor Warburg Pincus, in a deal valued at $4.25bn in cash. The business will offer services from clinical research to commercial deployment for clients.

MedCity News
OCTOBER 10, 2023
Bayer opens its new cell therapy manufacturing facility as it prepares a Parkinson’s disease cell therapy for Phase 2 testing. The new 100,000 square foot facility also has space for manufacturing other cell therapies in the pharma giant’s pipeline.

European Pharmaceutical Review
JULY 18, 2023
A market report by Meticulous Research has reported that rising pressure to reduce overall process costs has meant many pharma and biotech manufacturers are focusing on streamlining their processes by using quality management systems. What is driving adoption of quality management systems in pharma? billion by 2030.

European Pharmaceutical Review
SEPTEMBER 12, 2023
As part of The Future of Pharma/Biopharma Analysis 2023 , which took place on 28-29 June 2023, European Pharmaceutical Review ( EPR ) gathered an expert panel to discuss developing a modern contamination control strategy. Opening the session, moderator Tim Sandle highlighted six fundamental aspects of contamination control.
MedCity News
AUGUST 3, 2022
A panel discussion from a recent Microsoft summit offered insights on how tech companies are closely collaborating with the pharma industry as they seek to repair and improve manufacturing supply chain issues exposed by the Covid-19 pandemic.

pharmaphorum
DECEMBER 18, 2022
I outline below the salient aspects of a technology-driven patient access model, embraced as design principles, while designing and iterating a patient access platform with several large and small biopharma brands. The post Access solutions: the missing link that prevents biopharma innovation from reaching patients appeared first on.

Pharmaceutical Technology
JUNE 15, 2023
AI in pharma R&D is one of the latest innovations helping teams apply existing knowledge to new problems. Artificial intelligence (AI) has many implications for pharmaceutical companies, including on research, drug discovery and development, manufacturing and trials. Here are a few ways AI is used in pharma today.
MedCity News
APRIL 13, 2023
Rejection of Eli Lilly’s mirikizumab means that two of the four drugs the pharma giant identified as revenue growth drivers for 2023 have failed to pass the regulatory bar. One of those drugs has been approved while the fourth drug still awaits an FDA decision.

Clarivate
JULY 24, 2023
Biopharma and medtech brands made up a dozen of the Clarivate Top 100 New Brands for 2023, as the life science industries continued to show a muscular approach to protecting their intellectual property. The post Pharmas and medtechs flex brand protection among Top 100 New Brands for 2023 appeared first on Clarivate.

Pharmaceutical Technology
SEPTEMBER 8, 2022
The speciality drug channel participants, including speciality pharmacies, biopharma companies, and distributors, are transforming their operations with automated and secure data systems and next-generation analytics, which will improve data collection, partner performance and strategy effectiveness. Payer and/or manufacturer reporting.

MedCity News
JULY 6, 2022
Emergn Principal Consultant Mario Moreira shared insights on how pharma companies can use the principles of Value, Flow and Quality to shorten drug development timelines and deliver better patient outcomes through improved ways of working.

pharmaphorum
AUGUST 4, 2022
This is due to the specialist nature of roles and the growth and diversification of biopharma, which requires an increasingly sophisticated and skilled workforce. Furthermore, by simulating real-life scenarios, AR offers the chance for staff in the pharma industry to practice complex operations without the associated risks.

Pharmaceutical Technology
SEPTEMBER 13, 2022
The acquisition will expand the international solutions portfolio of AmerisourceBergen aiding in advancing its position as a preferred partner for biopharmaceutical manufacturers. This approach will support its manufacturer partners throughout the pharmaceutical development and marketing processes.

PM360
DECEMBER 20, 2023
With 2024 on the horizon, many in the biopharma space are taking stock of 2023 and what the current environment means for their potential success next year. This may best be achieved by large pharma companies, with their combined $200bn annual R&D budget, compared to smaller companies that are relying on external investment.

MedCity News
SEPTEMBER 24, 2023
Syntax Bio’s technology platform uses CRISPR to direct stem cells to become a desired cell type in a process that’s more scalable and less expensive than current methods. The startup is raising a Series A round of financing to further demonstrate the potential of its technology.

Pharmaceutical Technology
JULY 6, 2022
Just last week, trading for 4D Pharma , a British biotech, was suspended on the London Stock Exchange’s Alternative Investment Market , and the company will be delisted from NASDAQ on July 7. Other businesses such as Redhill Biopharma or Vincerx Pharma have also announced plans for large layoffs. Caution in the near future.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content