THINK Surgical’s TMINI System Receives FDA Special 510(k) Clearance for Use With Additional Implants
Legacy MEDSearch
OCTOBER 27, 2023
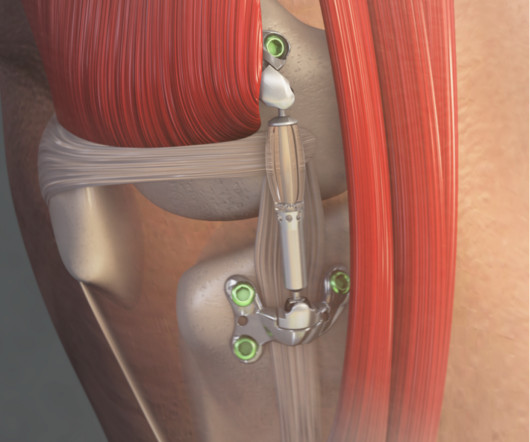
an innovator in the field of orthopedic surgical robots, today announced that its TMINI Miniature Robotic System has received Special 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use with implants from three additional implant manufacturers. THINK Surgical, Inc., ” About THINK Surgical, Inc.



















Let's personalize your content