Modifier gene therapy – clinical development and manufacturing considerations
European Pharmaceutical Review
JUNE 13, 2024
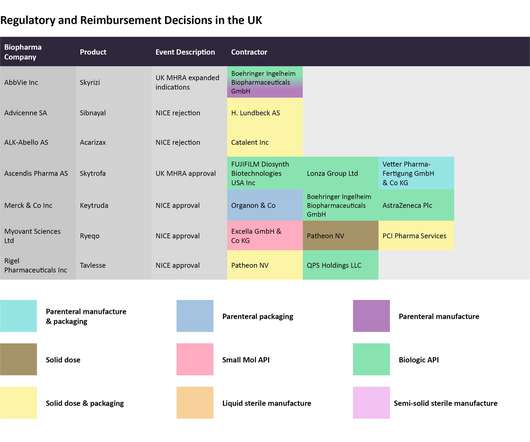
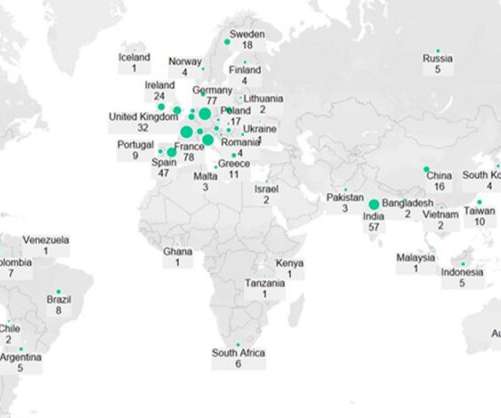
In April 2024, Ocugen received US Food and Drug Administration (FDA) clearance to initiate the Phase III liMeliGhT clinical trial for OCU400 for retinitis pigmentosa. In December 2023, the FDA granted OCU400 Regenerative Medicine Advanced Therapy (RMAT) designation. x 10 10 vg/eye of OCU400) and the untreated control group.



































Let's personalize your content