Abliva’s NV354 receives ODD from US FDA
Pharma Leaders
APRIL 17, 2023
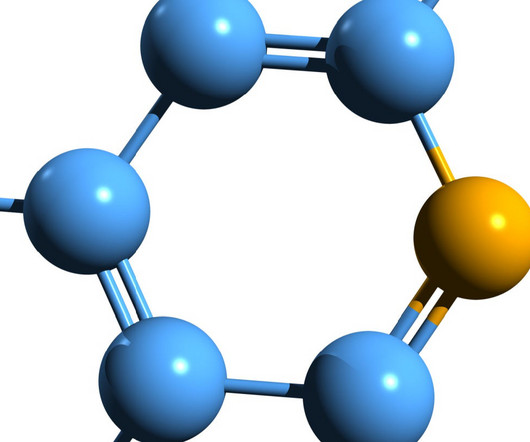
Sweden-based Abliva has announced that its drug candidate NV354 has been received orphan drug designation (ODD) from the US FDA Office of Orphan Products Development to treat mitochondrial disease. FDA is a validation of the quality of the NV354 pre-clinical program and another important milestone for Abliva.



















Let's personalize your content