Moderna's omicron-busting COVID-19 booster holds its own against latest variant BQ.1.1
Fierce Pharma
NOVEMBER 14, 2022
Moderna's omicron-busting COVID-19 booster holds its own against latest variant BQ.1.1. fkansteiner. Mon, 11/14/2022 - 11:11.

Fierce Pharma
NOVEMBER 14, 2022
Moderna's omicron-busting COVID-19 booster holds its own against latest variant BQ.1.1. fkansteiner. Mon, 11/14/2022 - 11:11.

Pharmaceutical Technology
NOVEMBER 14, 2022
In a field dominated by antibodies and small molecules, two cell-therapy based approaches have come under the spotlight for showing early signs of efficacy in treating lupus. In September, a group from Friedrich Alexander University Erlangen-Nuremberg reported that five patients with lupus achieved remission after an infusion of autologous chimeric antigen receptor (CAR)-T cells led to a deep depletion of B cells.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
NOVEMBER 14, 2022
AstraZeneca and Sanofi win big as Europe's human medicines committee endorses approvals, label expansions. fkansteiner. Mon, 11/14/2022 - 08:59.

MedCity News
NOVEMBER 14, 2022
A panel of healthcare data experts recently broke down some of healthcare’s most common buzzwords at HLTH. While the experts agreed that many industry professionals still struggle to grasp the true denotations of words and phrases like “interoperability” and “reimagining healthcare,” the panelists said these terms can be quite meaningful once they’re unpacked.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Fierce Pharma
NOVEMBER 14, 2022
As COVID vaccine market slows, AstraZeneca, Merck, Takeda and more get their chance to shine in Q3. fkansteiner. Mon, 11/14/2022 - 13:31.

MedCity News
NOVEMBER 14, 2022
GSK is making greater use of artificial intelligence and computational techniques as part of its drug discovery efforts. Speaking during the HLTH conference in Las Vegas, executive Mike Montello likened his company’s techniques to the efforts to study and analyze images from outer space.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

pharmaphorum
NOVEMBER 14, 2022
Sanofi and Regeneron’s Dupixent is on track to become the first targeted drug in the EU to be approved for prurigo nodularis (PN), a rare and highly debilitating skin disease. The EMA’s human medicines committee (CHMP) has recommended IL-4 and IL-13 inhibitor Dupixent (dupilumab) to treat adults with moderate to severe PN, a disease which causes hard lumps to form on the skin that are so itchy they can lead patients to scratch themselves to the point of bleeding or pain.

Fierce Pharma
NOVEMBER 14, 2022
In Jazz case, FTC urges court to delist patent on blockbuster Xyrem. esagonowsky. Mon, 11/14/2022 - 08:54.
Pharmaceutical Technology
NOVEMBER 14, 2022
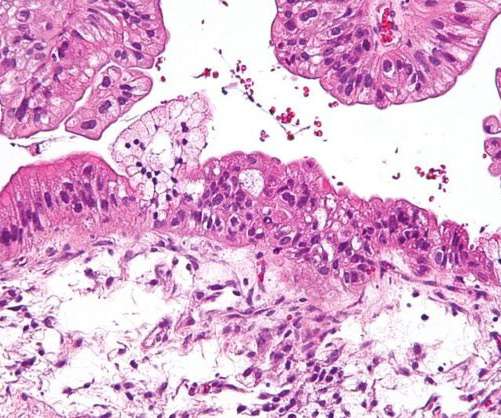
The US Food and Drug Administration (FDA) has granted accelerated approval for ImmunoGen’s Elahere (mirvetuximab soravtansine-gynx) to treat adults with folate receptor alpha (FR?)-positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer. The antibody-drug conjugate (ADC) treatment is indicated for such patients who have previously received one to three systemic therapy regimens, irrespective of the usage of Avastin priorly.

MedCity News
NOVEMBER 14, 2022
The new platform is a “digital front door” for members, offered on the web and through the My Highmark app. It will help members with scheduling appointments, accessing primary care and urgent care, managing prescription drugs and other services. It will roll out in January.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
pharmaphorum
NOVEMBER 14, 2022
Every fraction of a degree matters. COP27 is in full swing and world leaders are contending with the fact that we will likely not meet the Paris Agreement’s 1.5°C target. The reality is that we will need to focus on limiting a temperature rise wherever possible and that every fraction of a degree has a real and major consequence on our global environment.
MedCity News
NOVEMBER 14, 2022
Manual processes and inadequate analytics frequently prevent hospitals from achieving OR revenue and utilization goals. By using automation, behavioral science, and comprehensive real-world data, hospitals can unleash the full potential of their OR enterprises and supercharge perioperative growth.

PharmaVoice
NOVEMBER 14, 2022
On the surface, the Inflation Reduction Act is designed to lower drug prices, but its impact on innovation incentives could produce a landscape of new medicines that are costlier for patients.

MedCity News
NOVEMBER 14, 2022
Phone calls account for 70% of people’s first point of contact with a health system, so hospitals have sought to implement software that balances the need for speedy automation with intelligent systems that anticipate and best serve patient and care giver needs.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
Pharma Pathway
NOVEMBER 14, 2022
Ipca Laboratories Ltd- Openings for Freshers in Production/ Stores Dept. -Apply Now. Job Description. Openings for Freshers at Ipca -Piparia. Ipca Laboratories Ltd- Openings for Freshers in Production/ Stores Dept. -Apply Now. Department: Production/ Stores. Posts: Asst. Technician. Qualification: ITT/ HSC/ SSC. Experience: Freshers. Age: 18 to 23 years.

MedCity News
NOVEMBER 14, 2022
Cloud-based data management solutions help the industry meet its business and compliance challenges. These platforms need to replace traditional data management methods and workflows for biopharma companies and contract development and manufacturing organizations that seek competitive advantages.

European Pharmaceutical Review
NOVEMBER 14, 2022
Four medicines recommended for approval in the European Medicines Agency (EMA)’s Committee for Medicinal Products for Human Use (CHMP)’s latest meeting included a biosimilar for osteoporosis and a novel COVID-19 vaccine. VidPrevtyn Beta, recommended as a new recombinant, adjuvanted booster COVID-19 vaccine in adults previously vaccinated with an mRNA or adenoviral vector COVID-19 vaccine, is the seventh COVID-19 vaccine recommended for approval in the European Union (EU).

MedCity News
NOVEMBER 14, 2022
There are three major tensions that still need to be addressed in virtual care or telemedicine before it can work as promised. Convenient.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

Copyright Clearance Center
NOVEMBER 14, 2022
The post Cader Unravels Judge Pan’s Ruling To Block PRH/S&S Merger appeared first on Copyright Clearance Center.

MedCity News
NOVEMBER 14, 2022
Ex-Theranos CEO Elizabeth Holmes will face sentencing this week, and has asked the court to […].

PharmaVoice
NOVEMBER 14, 2022
An IQVIA Institute for Human Data Science report outlines how companies can improve communication, recruitment, data and investing strategies to accelerate clinical trial timelines.

Pharmaceutical Technology
NOVEMBER 14, 2022
BioNTech’s Singapore affiliate BioNTech Pharmaceuticals Asia Pacific has signed an agreement with Novartis Singapore Pharmaceutical Manufacturing to acquire a GMP-certified manufacturing site in the country. . The latest development is part of the expansion plans of BioNTech to boost its international footprint in Asia. . Backed by the Singapore Economic Development Board (EDB), the site will become completely operational late next year.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

Copyright Clearance Center
NOVEMBER 14, 2022
The post Is there a Crisis of Confidence in Scientific Research? appeared first on Copyright Clearance Center.

PharmaTimes
NOVEMBER 14, 2022
Data revealed that 100% of patients achieved a complete remission with no recurrence
Pharmacy Times
NOVEMBER 14, 2022
Despite evidence of a higher risk of myocarditis or pericarditis with COVID-19 vaccination, immunization should still be recommended because the benefits likely outweigh the harms, study shows.

PharmaTimes
NOVEMBER 14, 2022
Therapy is well-tolerated in allogeneic haematopoietic cell transplantation (HCT) patients

Advertisement
Clinical development organizations face a wide array of challenges when it comes to data, many of which can impact the operational effectiveness of their clinical trials. In this whitepaper, experts from Revvity Signals explore how solutions like TIBCO® Spotfire® enable better, more streamlined studies. The whitepaper also features a success story from Ambrx, a leading biopharmaceutical company, detailing how it has leveraged Spotfire to tackle data quality and collaboration challenges in clinic
Pharmaceutical Technology
NOVEMBER 14, 2022
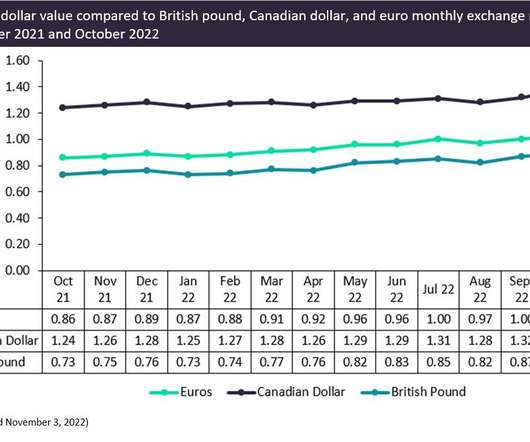
The UK pound has fallen against the US dollar consistently over the last year, causing many companies to suffer inflationary pressures and related hardships. Despite this, there is also a silver lining for British companies that export to countries like the US and others with currencies strengthening against the pound. Figure 1 shows the equivalent of one US dollar in British pounds, Canadian dollars, and euros on the 15th of each month for the last year.

PharmaTech
NOVEMBER 14, 2022
The new facility will allow ReiThera to consolidate its competitive position as a leading CDMO specialized in viral vector production.

Impetus Digital
NOVEMBER 14, 2022
Dr. Waqaas Al-Siddiq, Founder & CEO of Biotricity , sat down with me to discuss his journey from computer engineering to healthcare, innovation in the cardiac monitoring space, the concept of Mobile Cardiac Telemetry (MCT), and more. Here is a sneak peek of our conversation: Q: If you’re an individual that, let’s just say, for example, you have an arrhythmia, or you’ve had a congestive heart failure in the past and now need to be monitored.

PharmaTech
NOVEMBER 14, 2022
If you’re in the business of pharmaceutical product development or manufacturing for the US market, this brief talk by Dr. Richard Sidwell is for you. The need for pharmaceutical products is higher than ever. At the same time, the ability to develop and supply new products is being constrained by demographics, geopolitics, energy, and inflation. This is why it’s more important than ever to strategically invest in CMC development and engineering, onshoring, and thoughtful supply chain design.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content