CRO EastHORN picked up by Novotech
PharmaTimes
JANUARY 12, 2023
Agreement ensures that clients can access Novotech's early to late-phase CRO services across Europe
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 cro
cro 
PharmaTimes
JANUARY 12, 2023
Agreement ensures that clients can access Novotech's early to late-phase CRO services across Europe

PharmaVoice
FEBRUARY 28, 2024
From his advisory board role at Lindus Health, Robert Langer is aiming to overcome challenges in the CRO market.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharma Pathway
JANUARY 3, 2023
Sai Life Sciences Limited- Walk-In Drive for Analytical R&D for API & CRO Roles On 7th Jan’ 2023. Walk-In Interview for Analytical R&D for API & CRO Roles in Sai Life Sciences, Hyderabad. Department: Analytical R&D – API & CRO. Job Description. Qualification: M.Sc/ M.Pharm.

Pharma Pathway
SEPTEMBER 22, 2022
Aragen Life Sciences – Walk-In Interviews for Synthesis R&D (CRO) Dept. Walk-In Drive for Research Associate / Senior Research Associate/ Associate Scientist -Synthesis R&D (CRO) Dept. Experience: Synthesis R&D (CRO)-M.SC: 2 to 10 years/ Ph.D: On 24th Sept’ 2022. Job Description. 1 to 2 years/ PDF: 1 year.

Advertisement
When a CRO is bidding on a project where clinical supplies will be one of the aspects to manage on behalf of the client via a partner, leveraging the expertise of a chosen clinical supply partner can be a valuable resource in demonstrating the CRO’s understanding of and ability to deliver upon critical drug-supply related aspects of the project, and (..)

Spotio
FEBRUARY 21, 2023
Before SPOTIO, Scott was the CRO at Igloo and PowerDMS, where he was instrumental in developing the go-to-market strategies that delivered record ARR growth. He joined the team after serving as the CRO at ListenFirst, where he helped the company transition to financial stability.

Spotio
FEBRUARY 21, 2023
Before SPOTIO, Scott was the CRO at Igloo and PowerDMS, where he was instrumental in developing the go-to-market strategies that delivered record ARR growth. He joined the team after serving as the CRO at ListenFirst, where he helped the company transition to financial stability.

Fierce Pharma
NOVEMBER 3, 2023
Holding more than 25 years of experience in the medical sector spread across numerous pharmaceutical and CRO-based clinical research roles, Edel Shaw knows a thing or two about drug development.

PharmaTech
FEBRUARY 15, 2024
The need for preclinical testing expertise is growing as molecular complexity increases.

Pharmaceutical Technology
NOVEMBER 28, 2023
A Phase IIb/III trial studying the company’s immunotherapy izokibep, failed to achieve statistical significance for its primary endpoint

Pharma Leaders
AUGUST 4, 2022
Today, Validcare announces the integration of Real-World Research (RWR) Ltd into the Validcare Digital 1 st CRO platform. Denver, CO. August 2, 2022. RWR is a global leader in providing Real-World Evidence solutions to the pharmaceutical, biotech and medical device industries.

PharmaTech
JANUARY 31, 2023
Be the Match BioTherapies’ and CIBMTR’s expanded service offering is intended to aid in the design, implementation, and oversight of cellular therapy clinical trials.

Pharmaceutical Commerce
MAY 1, 2024
The CRO earned a Bronze rating for criteria based on environmental, social, and ethical performance.

PharmaVoice
FEBRUARY 27, 2023
Staffing shortages are causing inefficiencies in clinical trials, but one leading CRO is finding ways to adapt.

Pharmaceutical Technology
FEBRUARY 9, 2024
The survey by CRO Worldwide Clinical Trials shows that pharma and biotechs favour midsize CROs due to the personalised service.

European Pharmaceutical Review
JULY 5, 2023
The global clinical research organisation (CRO) market is expected to reach $139.56 Challenges for the CRO market Non-uniformity in regulations within various governments is expected to restrain the CRO market going forward, the research predicted. billion by the year 2033, a report by Future Market Insights has stated.

Pharmaceutical Technology
NOVEMBER 29, 2023
Closing the first day of the CTO Europe conference, a global CRO shares insights on the drug development lifecycle in oncology.

PharmaVoice
AUGUST 31, 2022
Fresh off an eight-year tour of duty at ICON, the CRO industry veteran is stepping into a new role and a new title as chief delivery officer at Thread.

Pharma Pathway
SEPTEMBER 16, 2022
Roles: Senior Research Associate; CRO -R&D. Research Scientist- CRO- R&D. 2-10 years of relevant experience with CRO Industry. Job Location: Ahmedabad, Gujarat- CRO-R&D (Medchem). Qualification & Experience: M.Sc (Organic Chemistry), M.Pharm (Pharmaceuticals Chemistry/ Medicinal Chemistry) , P.h.D

PharmaTech
DECEMBER 2, 2023
The rising complexity of clinical research protocols has necessarily expanded the CRO landscape.

Pharma Pathway
SEPTEMBER 22, 2022
Roles: Senior Research Associate; CRO -R&D. Research Scientist- CRO- R&D. 2-10 years of relevant experience with CRO Industry. Job Location: Ahmedabad, Gujarat- CRO-R&D (Medchem). Eligible Criteria: . Organic Chemistry/ Natural Product Synthesis). Fresh PhDs or Post Docs are also eligible.
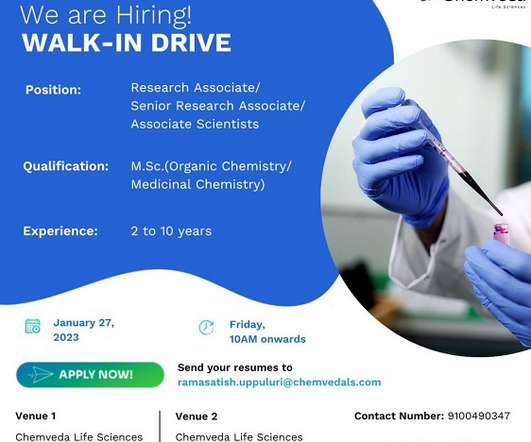
Pharma Pathway
JANUARY 24, 2023
Ltd- Walk-In Interviews for Discovery Chemistry Solutions On 27th Jan’ 2023 Job Description Dear Candidate, We are conducting Walk-In Interviews for multiple positions in Discovery Chemistry Solutions department (CRO) @ Chemveda Life Sciences Pvt. Organic chemistry/ Medicinal Chemistry) with 1-07 years relevant CRO Exp.

European Pharmaceutical Review
JANUARY 24, 2023
Previously, Maria worked as a senior clinical research physician for Parexel, a contract research organisation (CRO) in Phase I trials in Berlin. This will help to maximise on cross-selling opportunities, providing a seamless and customer-centric experience to Velocity’s pharma and CRO partners.

PharmaVoice
APRIL 17, 2023
Gain a deep understanding of the analytical challenges that gene therapy developers face, and how a strategic partnership with a specialized CRO can offer a competitive advantage in accelerating timelines and improving program success.

Pharmaceutical Technology
JULY 4, 2023
The spin-off will create a new global Phase I-IV CRO, patient access and technology solutions.

European Pharmaceutical Review
OCTOBER 11, 2022
The study produced by the University of Cambridge has indicated that pharmaceutical companies are increasingly leaning away from one-time transactions with contract research organisations (CRO), to preferred providers and multi-year strategic partnerships. Thus, outsourcing development to CROs has become a popular cost-effective solution.
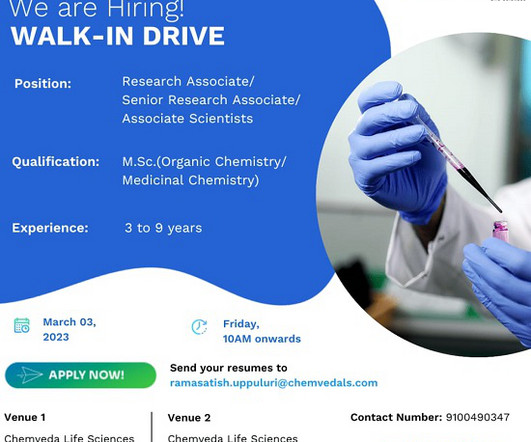
Pharma Pathway
MARCH 2, 2023
Ltd- Walk-In Interviews for Discovery Chemistry Solutions On 3rd Mar’ 2023 Job Description Dear Candidate, We are conducting Walk-In Interviews for multiple positions in Discovery Chemistry Solutions department (CRO) @ Chemveda Life Sciences Pvt. Organic chemistry/ Medicinal Chemistry) with 3-09 years relevant CRO Exp.

Pharma Pathway
MARCH 4, 2023
Ideal Candidates should be highly organized with the ability to work independently in a fast-paced team environment in a transparent and entrepreneur culture We are conducting Walk-In Interviews at Ahmedabad. Please refer to the creative for venue details and timings.

PharmaTech
JUNE 4, 2023
Our imaging consultants can serve as an intermediary between you and and an imaging CRO to launch a complete and accomplished clinical trial. At Bracken, our experts bring storied experience from the intersection of medical imaging and clinical research.

Pharmaceutical Technology
JANUARY 11, 2023
Avance Clinical is a full-service contract research organisation (CRO) that provides clinical research capabilities for biotech companies worldwide. The company specialises in offering high-touch clinical trial solutions tailored to clients’ individual needs.

Pharma Pathway
DECEMBER 17, 2022
Should be from Core Pharma or Cro or Pharma clients IT shared Services Industrial Expertise. We have Opportunities in our Clinical Data Management division for people who have the below mentioned exposure: Skillset – End to end CDM with Rave or Veeva exposure. Experience: 03 to 06 years. Notice Period: Any.

European Pharmaceutical Review
NOVEMBER 1, 2022
Additionally, the parked PE capital will support strong contract sector valuations and, more significantly, investment in CRO/CDMO infrastructure. percent 1 for CROs and 8.5 percent 1 for CROs and 8.5 Significantly, Scanlan states that the current industry-predicted growth rates of 7.2

PharmaTech
SEPTEMBER 9, 2022
Avance Clinical’s acquisition of C3 Research Associates expands its CRO services to North America.

Pharma Pathway
SEPTEMBER 16, 2022
We are looking for candidates with 1-5 years of work experience in the CRO/ CRAMS/ API Development Documentation Team. Ltd-Walk-In Interview for Quality Control/ Analytical R&D On 17th Sept’ 2022. Job Description. For Quality Control/ Analytical R&D . Departments: QC/ AR&D. Qualification: M.Sc. Experience: 01 to 05 years.

Pharma Pathway
DECEMBER 7, 2022
Dear Candidate, We are conducting Walk-In Interviews for multiple positions in Discovery Chemistry Solutions department (CRO) @ Chemveda Life Sciences Pvt. Organic chemistry/ Medicinal Chemistry) with 1-7 years relevant CRO Exp. Department: Discovery Chemistry Solutions. Designation: Research Associate/ Sr. Qualification: M.Sc.

Pharma Leaders
MAY 12, 2023
Founded in 2001 and headquartered in Guelph, Ontario, Canada, Nutrasource is a global contract research organisation (CRO) in the pharmaceutical and nutraceutical businesses. The company will initially buy a 60% interest in Nutrasource while having the right to pick the remaining 40% holding in 2026.

Pharma Pathway
JANUARY 27, 2023
Ltd Department: R&D- Synthesis / Process/ CRO-API (Males) Qualification: M.Sc Experience: 01 to 04 years Department: AR&D- Method Validation API (Males) Qualification: M.Sc/ M.

Pharma Leaders
JUNE 12, 2023
Private equity and growth capital firm EdgeCap Partners has acquired a majority stake in Bulgarian clinical research solutions organisation (CRO) Comac Medical. Comac founder and CEO Dr Milen Vrabevski and its other shareholders will retain a significant minority stake. The companies have not disclosed the financial terms of the deal.

Pharma Pathway
DECEMBER 14, 2022
Department: R&D- (Synthesis / Process/ CRO). Walk-In Interview for R&D/AR&D/ Regulatory Affairs/ IPM/ FR&D/ FAR&D On 15th & 16th Dec’ 2022 @ Biophore India Pharmaceuticals Pvt. Qualification: M.Sc. Experience: 01 to 04 years. Department: AR&D- (Method Validation). Qualification: M.Sc/ M.
pharmaphorum
NOVEMBER 20, 2022
In fact, an established contract research organisation (CRO) will play an active role in setting immunogenicity assay standards that are used globally. When coupled with transparency, the agility of a CRO, and when grounded in science, a vaccine can quickly demonstrate its global potential in tackling public health challenges.

World of DTC Marketing
FEBRUARY 10, 2022
Of the 16 states with adult obesity rates 35 percent or higher, 10 — Alabama, Arkansas, Indiana, Kentucky, Louisiana, Mississippi, Ohio, Oklahoma, Tennessee and Texas — rank in the top 20 for age-adjusted per capita covid death rates, according to Bioinformatics CRO, a consulting firm. Washington Post.

Pharmaceutical Technology
MAY 18, 2023
Gate Neuroscience has already identified the CRO for the trial alongside some initial trial sites, he says. The company currently aims to study the drug in a group of 160 subjects, which will be evenly split between those on the drug and on placebo, says Houghtaling. The trial will take place at US clinical sites, adds Houghtaling.

European Pharmaceutical Review
APRIL 5, 2023
Key developments in the market In Jan 2023, a new strategic partnership was announced between a contract research organisation (CRO) and a supply chain solutions firm. Other obstacles in this market are the provision of personalised treatments and smart packaging, the research stated.
Pharmaceutical Technology
NOVEMBER 4, 2022
Reaction Biology CEO John Johnson said: “vivoPharm’s US operations are highly complementary to Reaction’s existing suite of drug discovery CRO services, enabling us to build upon our legacy of excellent science and customer-centric innovation.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content