Krystal Biotech touts topical gene therapy uptake, plots overseas launches
Fierce Pharma
MAY 7, 2024
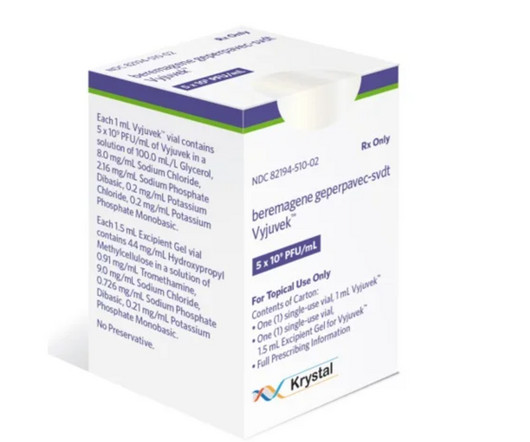
After a 2023 approval from the FDA, Krystal Biotech has collected more than $95 million from its launch of Vyjuvek, the first treatment for the rare skin disease dystrophic epidermolysis bullo | The topical gene therapy has pulled in nearly $100 million during its first three quarters on the U.S.















































Let's personalize your content