Are timely access and robust safety mutually exclusive?
pharmaphorum
JULY 6, 2022
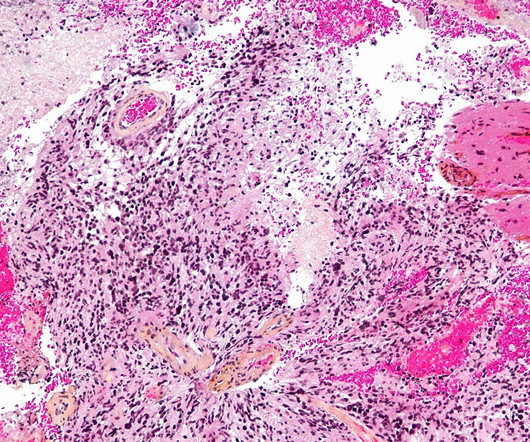
The FDA approves new cancer treatments in half the time of the EMA – but does faster mean better? And how can regulators balance timely access with robust safety? Cancer patients in Europe wait an average of almost eight months longer for access to breakthrough medications than their American counterparts.




































Let's personalize your content