In Equal Measures: The Importance of Excipient Quality
PharmaTech
SEPTEMBER 2, 2023
Excipients should be treated equally when it comes to quality management, risk assessment, and testing.
PharmaTech
SEPTEMBER 2, 2023
Excipients should be treated equally when it comes to quality management, risk assessment, and testing.

PharmaTech
SEPTEMBER 2, 2023
Coprocessed excipients save time and cost while improving performance in a widening array of dosage forms.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PharmaTech
SEPTEMBER 2, 2023
Audits and inspections both assure that requirements have been fulfilled and whether documented proof is available, says Siegfried Schmitt, vice president, Technical at Parexel.

PharmaTech
SEPTEMBER 2, 2023
The ROSS line of Double Planetary Mixers are fully customized and engineered for efficient processing of medical-grade silicone formulations in a portable workstation.
PharmaTech
SEPTEMBER 2, 2023
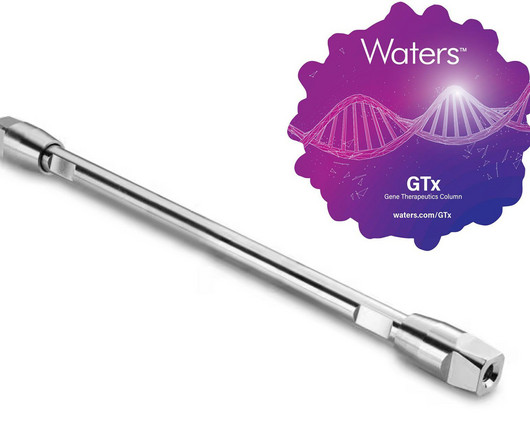
Waters Corporation’s XBridge Premier GTx BEH size exclusion chromatography (SEC) columns are designed to improve analysis while lowering the cost of gene therapies.

PharmaTech
SEPTEMBER 2, 2023
Recent research and perspectives shed light on an opportunity to better connect risk and knowledge through improved integration of systems.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

PharmaTech
SEPTEMBER 2, 2023
The ACT EU initiative aims to develop the European Union further as a competitive centre for innovative clinical research.

PharmaTech
SEPTEMBER 2, 2023
Pharma's ability to continually reinvent itself will be critical in growing future business operations.

PharmaTech
SEPTEMBER 2, 2023
There are many promising trials in the pipeline that may bring big news for major therapeutic areas.

PharmaTech
SEPTEMBER 2, 2023
A structured cleaning procedure is essential to ensure the effective cleaning of equipment.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

PharmaTech
SEPTEMBER 2, 2023
FDA is anticipating how AI may advance manufacturing and improve supply chain security.
PharmaTech
SEPTEMBER 2, 2023
Tumour-infiltrating lymphocyte (TIL) therapies offer a new route to target cancer.

PharmaTech
SEPTEMBER 2, 2023
European vaccine manufacturing is being boosted with high level investments.

PharmaTech
SEPTEMBER 2, 2023
Modular facilities allow for a certain amount of flexibility and reconfiguration options.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
Let's personalize your content