Editas grabs orphan drug status for sickle cell disease CRISPR therapy
Pharmaceutical Technology
APRIL 28, 2023
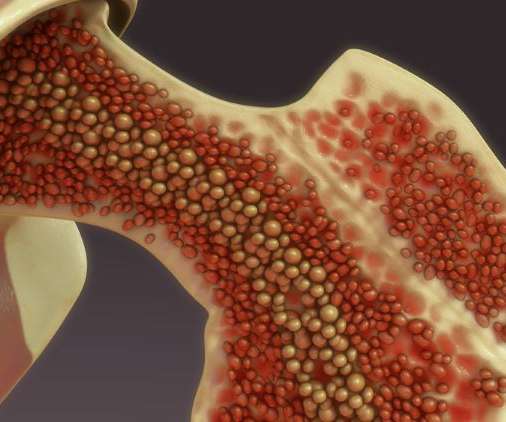
The US Food and Drug Administration (FDA) has granted an Orphan Drug Designation to Editas Medicine’s gene therapy EDIT-301 in sickle cell disease, based on an April 27 announcement. The US agency previously granted the Orphan Drug Designation to EDIT-301 for its study in beta thalassemia, in May 2022.
















Let's personalize your content