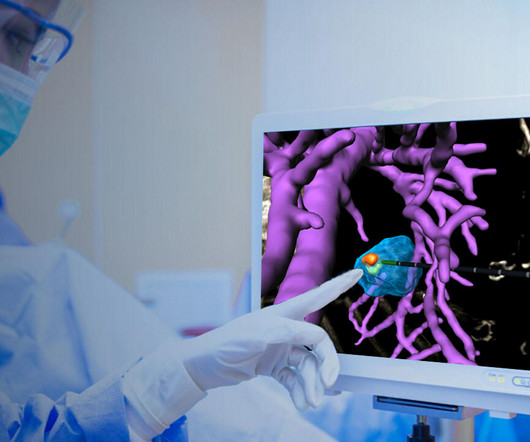
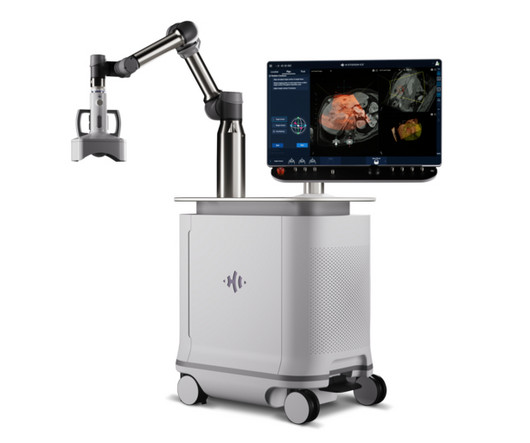
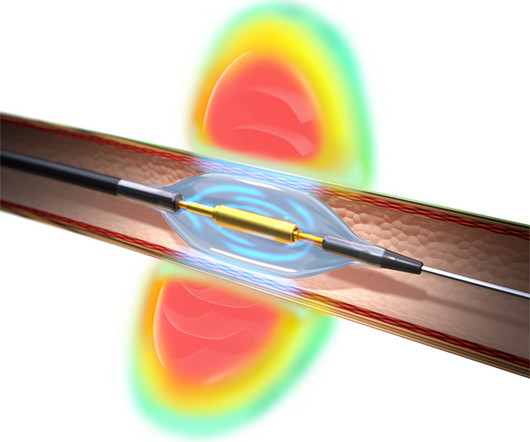
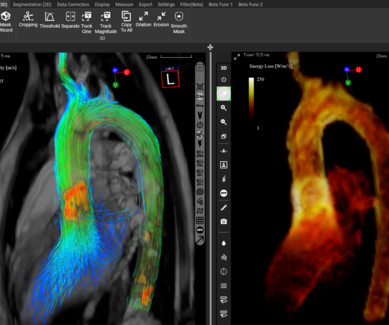
Techsomed Announces FDA Clearance for Ablation Treatment Planning and Confirmation Software
Legacy MEDSearch
SEPTEMBER 5, 2023
a medical software innovator dedicated to enhancing clinical impact in ablation therapy, announced today that it has received 510(k) clearance from the USA Food and Drug Administration (FDA) for its VisAble.IO solution is not yet approved for sale or distribution in the USA and is limited by USA law to investigational use.














Let's personalize your content