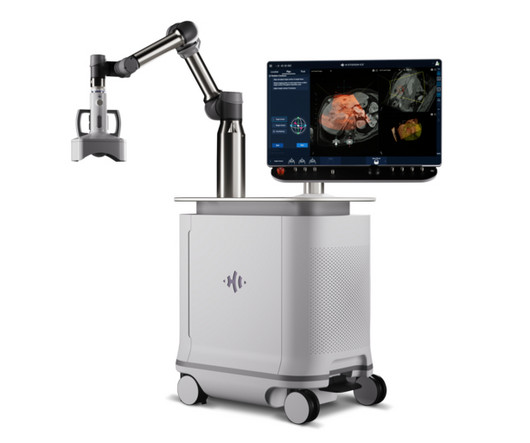
FDA Awards HistoSonics Clearance of its First-of-a-Kind Edison® Histotripsy System
Legacy MEDSearch
OCTOBER 9, 2023
Food and Drug Administration’s (FDA) De Novo Classification Request process, a rigorous pre-market review pathway for medical devices with no existing predicate. Marketing authorization makes Edison the first and only histotripsy platform available in the Unites States.









Let's personalize your content