Cybin bags breakthrough tag for psychedelic depression drug
pharmaphorum
MARCH 14, 2024
Cybin files a $150m placement on the back of new clinical data with its psychedelic drug CYB003 for depression and an FDA breakthrough designation.

 tag depression
tag depression 
pharmaphorum
MARCH 14, 2024
Cybin files a $150m placement on the back of new clinical data with its psychedelic drug CYB003 for depression and an FDA breakthrough designation.

pharmaphorum
AUGUST 21, 2022
Shares in Axsome Therapeutics have rocketed on FDA approval of its depression therapy Auvelity (formerly AXS-05) – a year after its approval was held up by the regulator. The post Axsome bounces back as FDA clears depression drug appeared first on. Photo by Sydney Sims on Unsplash.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
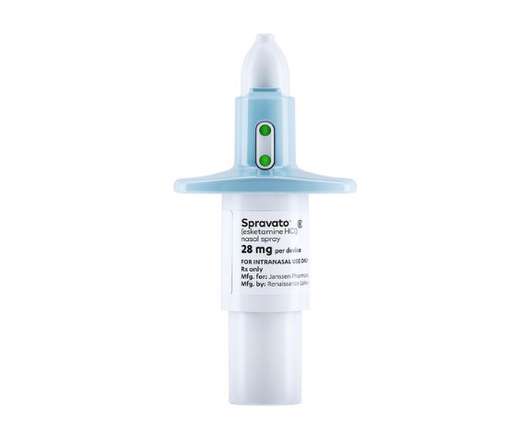
pharmaphorum
NOVEMBER 24, 2022
Johnson & Johnson’s Spravato therapy for treatment-resistant depression (TRD) hasn’t made the impact the company hoped for in the market, and it is looking to new clinical results to accelerate its take-up. Both drugs were given on top of standard antidepressants to patients wo had failed a least two earlier treatments.

Pharma Marketing Network
JUNE 8, 2020
We are now in a 20-yearlong economic depression for the working and middle class as a result of health care prices inflating, prices not cost. Dave (HR): Yeah, I mean really everything we’re doing you know is about overturning the devastating effects of the status quo health plans. So that individuals can realize their full potential.
Let's personalize your content