Cancer: Progress but a long way to go
World of DTC Marketing
MARCH 17, 2022
In recent years, early detection and treatment improvements have helped boost the 3-year survival rate for lung cancer from 21% in 2004 to 31% in 2015 through 2017. But it’s’ still the leading cause of cancer deaths. The Food and Drug Administration approved them, an analysis JAMA Internal Medicine found.

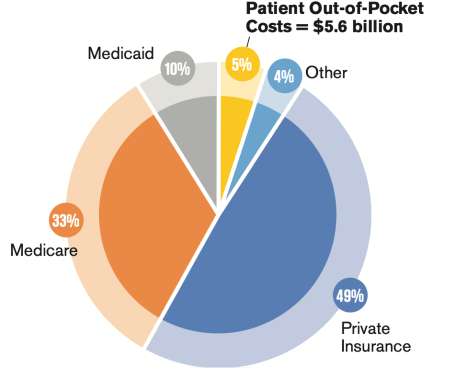







Let's personalize your content