New Partnership Brings ‘Food Is Medicine’ Programs to Veterans
MedCity News
APRIL 28, 2023
The Rockefeller Foundation is funding two produce prescription pilot programs at VA healthcare systems in Salt Lake City, Utah, and Houston, Texas.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

MedCity News
APRIL 28, 2023
The Rockefeller Foundation is funding two produce prescription pilot programs at VA healthcare systems in Salt Lake City, Utah, and Houston, Texas.

PharmExec
NOVEMBER 9, 2023
Healthy Food Rx provides home-delivered medical prescriptions of healthy food to help address diabetes.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

MedCity News
FEBRUARY 2, 2024
Foodsmart is rolling out its new program that enables doctors to refer their patients to Foodsmart’s dieticians and healthy eating platform, as well as write prescriptions for healthy food just as easily as they might a normal pharmacy prescription.

Pharmaceutical Technology
OCTOBER 13, 2022
Melatonin has a prescription-only status and is strictly regulated in the UK, making this the first time it has been approved for use in this indication. Colonis then sent a corrective letter to the recipients, but a Prescription Medicines Code of Practice Authority panel pointed out that it didn’t specify this was required by the MHRA.
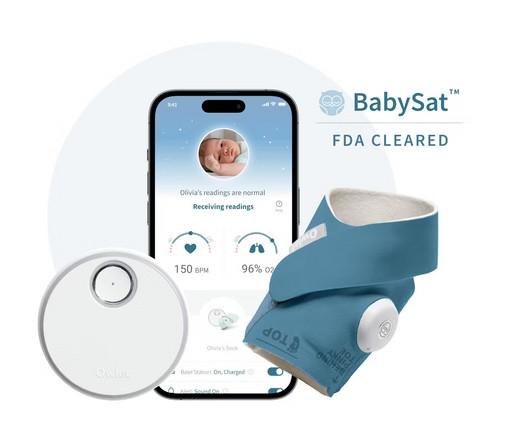
Legacy MEDSearch
JUNE 20, 2023
Food and Drug Administration (“FDA”) of BabySat , the first medical pulse-oximetry device featuring its advanced, wire-free sock design. The post Owlet Announces FDA-Clearance of the First Prescription Pulse Oximetry Sock for Infants appeared first on Legacy MedSearch | Medical Device Recruiters. Are you hiring?

Pharmacy Times
AUGUST 13, 2023
Pharmacists can improve patient access to both healthful foods and necessary medicines as a continuum of care in underserved communities.
World of DTC Marketing
JANUARY 7, 2022
When they say compare prescription drug prices, they mean the cost of a prescription if you aren’t using insurance. When you use GoodRx, ALL that data regarding your prescription transaction is collected and stored. Right now, there’s a lot of investment hype around Food Rx. And advertising?
Pharmaceutical Technology
MAY 19, 2023
The US Food and Drug Administration (FDA) has granted approval to Bausch + Lomb and Novaliq’s Miebo (perfluorohexyloctane ophthalmic solution) to treat the signs and symptoms of dry eye disease (DED). It is the first and only prescription eye drop to receive FDA approval to treat DED that targets tear evaporation directly.

World of DTC Marketing
FEBRUARY 1, 2021
SUMMARY: The biggest argument against pharma is the cost of prescription drugs. The most common complaint against the pharma industry is the high cost of some prescription drugs. The media usually runs a story about someone who lost their insurance and has to choose between their medications or putting food on the table.
World of DTC Marketing
JULY 17, 2022
Unpaid celebrity endorsements include venture capitalist Marc Andreessen, who says the drug completely changed his relationship with food, and Elon Musk, who has cited it on Twitter. Payers also face hurdles, so patients must obtain permission before filling a prescription. So far, the data for Wegovy looks excellent.

World of DTC Marketing
DECEMBER 2, 2021
Ad recall is essential for CPGs, but for prescription drugs, the question that’s missing is “what action did it lead to? Consumers often don’t make rational choices when it comes to products, including prescription drugs. ” Uh…no. The post A scientific approach to irrational consumer choices.

World of DTC Marketing
APRIL 12, 2021
The US Food and Drug Administration (FDA) believes the adulteration of heparin was an economically motivated act—a clear breach of the US pharmaceutical supply chain. The answer lies in both an outdated way we develop prescription drugs and the continued quest for the next big blockbuster. In other words, money over safety.

Pharmaceutical Technology
JUNE 21, 2023
AGEPHA Pharma has received approval from the US Food and Drug Administration (FDA) for LODOCO (colchicine, 0.5 The company expects to launch LODOCO for prescription use in the second half of 2023. mg tablet) to treat cardiovascular disease.
Pharmaceutical Technology
MAY 26, 2023
The US Food and Drug Administration (FDA) has granted priority review for Takeda and HUTCHMED’s new drug application (NDA) for fruquintinib. The FDA has now assigned a prescription drug user fee act action date of 23 November 2023 for the NDA.
Pharmaceutical Technology
MAY 25, 2023
The US Food and Drug Administration has set 23 September 2023 as the prescription drug user fee action date for ATI-1501.

Legacy MEDSearch
MARCH 7, 2024
Food and Drug Administration (FDA) announced the clearance of Dexcom’s device, marking it as the first continuous glucose monitor available over the counter. Dexcom disclosed its plans to make Stelo available for purchase online without the need for a prescription, starting in summer 2024. On March 5, the U.S.

Pharmaceutical Technology
JUNE 12, 2023
Patients with erectile dysfunction (ED) will now be able to access a topical treatment without the need for a prescription after the US Food and Drug Administration (FDA) greenlit Futura Medical’s Eroxon. The addition of a topical gel to pharmacy shelves expands treatment options available to those with ED.

MedCity News
JUNE 13, 2023
They’ll be able to do this on the same platform they use to request non-emergency medical transportation and prescription delivery. Providers and payers will be able to use Uber Health to deliver healthy groceries and over-the-counter items to patients who need them.
Pharmaceutical Technology
MAY 2, 2023
The US Food and Drug Administration (FDA) has accepted the supplemental biologics licence application submitted by Bristol Myers Squibb for Reblozyl (luspatercept-aamt) as a first-line treatment of anaemia in adults with lower-risk myelodysplastic syndromes (MDS).
World of DTC Marketing
NOVEMBER 24, 2021
Food and Drug Administration rose to nearly $15,000 from about $6,000, the data showed. One-quarter of all cancer patients chose not to fill a prescription due to cost, according to a 2013 study in The Oncologist. From 2009-10 to 2018-19, the lowest average monthly costs for new cancer drugs approved by the U.S.
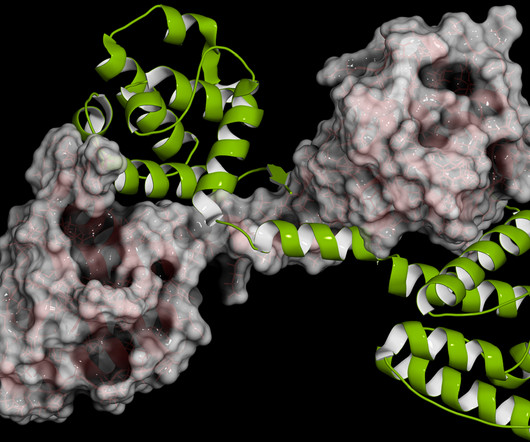
Pharmaceutical Technology
JUNE 20, 2023
The US Food and Drug Administration (FDA) has now granted fast-track and rare paediatric disease designations, and the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA) has also granted promising innovative medicine status to vamorolone for DMD.

Pharmaceutical Technology
JUNE 19, 2023
UK-based pharmaceutical giant GSK has announced that the US Food and Drug Administration (FDA) has extended the review period of its new drug application (NDA) for the rare bone cancer drug momelotinib by three months. The reason for this review extension was undisclosed in the announcement.

World of DTC Marketing
MAY 16, 2022
Food and Drug Administration approved Eli Lilly’s novel treatment for diabetes. When it comes to losing weight, Americans want shortcuts, and if it’s a prescription drug, it doesn’t matter. If You’re Short On Time: The U.S. The drug led to impressive blood sugar and body weight drops in clinical trials.
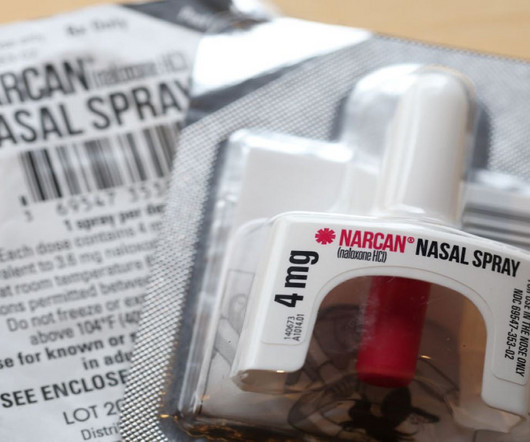
Pharmaceutical Technology
MARCH 30, 2023
On March 29, the US Food and Drug Administration (FDA) granted approval for the first nonprescription, “over-the-counter” (OTC) naloxone nasal spray, Narcan. Between 1999 and 2021, overdose deaths from synthetic opioids excluding methadone increased 97-fold, and prescription opioid overdose deaths increased 4.9-fold,

Pharmaceutical Technology
APRIL 14, 2023
Alvotech has received a complete response letter (CRL) from the US Food and Drug Administration (FDA) regarding its biologics licence application (BLA) for AVT02, a biosimilar to Humira (adalimumab).

Pharmaceutical Technology
MAY 16, 2023
In January 2023, the US Food and Drug Administration (FDA) approved lecanemab under the accelerated approval pathway to treat AD. The regulator has also accepted Eisai’s supplemental biologics licence application (sBLA) for lecanemab and granted priority review, with 6 July 2023 as the prescription drug user fee act (PDUFA) action date.

Pharmaceutical Technology
MAY 19, 2023
The US Food and Drug Administration (FDA) has accepted Satsuma Pharmaceuticals’ 505(b)(2) new drug application (NDA) for STS101 for acute treatment of migraine, for review. The prescription drug user fee act (PDUFA) date is expected to be set as January 2024.
Pharmaceutical Technology
OCTOBER 1, 2022
The US Food and Drug Administration (FDA) has granted approval for Amylyx Pharmaceuticals’ Relyvrio (sodium phenylbutyrate and taurursodiol) to treat adult patients with amyotrophic lateral sclerosis (ALS). A progressive and fatal neurodegenerative ailment, ALS is caused by the death of motor neurons in the brain and spinal cord.
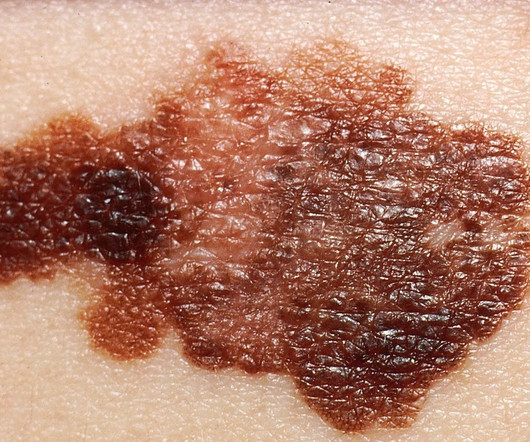
Pharmaceutical Technology
MAY 29, 2023
The US Food and Drug Administration (FDA) has accepted Iovance Biotherapeutics’ biologics license application (BLA) for lifileucel to treat advanced melanoma. It has been granted priority review by the FDA, with a target action date of 25 November this year, under the Prescription Drug User Fee Act (PDUFA).

Pharmaceutical Technology
MAY 5, 2023
The US Food and Drug Administration (FDA) has accepted Optinose’s supplemental new drug application (sNDA) for Xhance (fluticasone propionate) to treat chronic rhinosinusitis, which is now under review. The regulator has set 16 December 2023 as a prescription drug user fee act (PDUFA) target goal date.

World of DTC Marketing
SEPTEMBER 7, 2022
In pharma, growth depends on new products with hefty price tags when over 80% of voters want lower costs for their prescription drugs. Wall Street wants growth, as do investors, but that may be impossible for pharmaceutical companies. Biogen’s failure should be a warning to other pharma companies. ” Why are the costs so high?
Pharmaceutical Technology
JUNE 15, 2023
The US Food and Drug Administration (FDA) has accepted Ipsen’s supplemental new drug application (sNDA) for the Onivyde regimen as a first-line treatment for metastatic pancreatic ductal adenocarcinoma (mPDAC) patients. The regulator has set 13 February 2024 as a Prescription Drug User Fee Act goal date for the review of Ipsen’s sNDA.

Pharmaceutical Technology
JUNE 14, 2023
NOV03 is the first and only water-free and preservative-free prescription treatment for dry eye disease, and targets the evaporation of tears by forming a monolayer at the tear film’s air-liquid interface. Novaliq has also received US Food and Drug Administration approval for VEVYE (cyclosporine ophthalmic solution) 0.1%

Pharmaceutical Technology
JUNE 13, 2023
The US Food and Drug Administration (FDA) has granted approval for Ironwood Pharmaceuticals’ LINZESS (linaclotide) as a once-a-day treatment for functional constipation in paediatric patients aged between six and 17 years.

Pharmaceutical Technology
APRIL 21, 2023
The US Food and Drug Administration (FDA) has pushed quizartinib’s Prescription Drug User Fee Act (PDUFA) date in newly diagnosed FLT3-ITD positive acute myeloid leukemia (AML) for the review of updates of Risk Evaluation and Mitigation Strategies (REMS) included in the application.

World of DTC Marketing
JULY 5, 2022
Pharma websites provide the prescription that can help people overcome health problems but too often, they ignore the patient’s responsibility to lose weight and exercise. While the population is more worried about COVID, they are killing themselves by overeating processed food and not getting any exercise.
Pharma Leaders
JUNE 21, 2023
The US Food and Drug Administration (FDA) has accepted bluebird bio’s Biologics License Application (BLA) for lovotibeglogene autotemcel (lovo-cel) gene therapy and granted priority review for the product’s use in the treatment of sickle cell disease (SCD). We look forward to working with the agency on its review.”

Pharmaceutical Technology
JUNE 14, 2023
Ipsen has received US Food and Drug Administration (FDA) approval for Bylvay (odevixibat) to treat patients aged 12 months and above with cholestatic pruritus caused by Alagille syndrome (ALGS). The therapy is being offered immediately through prescription for eligible ALGS patients.

World of DTC Marketing
JUNE 22, 2022
A new feature—a sensor that monitors changes in the heart rate for irregularities that can lead to strokes and heart failure—has just been approved by America’s Food and Drug Administration (FDA). If pharmacies try to fill those generic prescriptions, the claims are rejected. Not if it affects their bottom line.

Pharmaceutical Technology
FEBRUARY 8, 2023
The US Food and Drug Administration (FDA) has accepted Aldeyra Therapeutics’ new drug application (NDA) for topical ocular reproxalap to treat signs and symptoms of dry eye disease. The regulator has assigned 23 November 2023 as the Prescription Drug User Fee Act (PDUFAs) date for its decision.

pharmaphorum
AUGUST 11, 2022
Zantac was originated by GSK and launched by the company in the early 1908s, which sold it as a prescription and over-the-counter (OTC) product for indications like heartburn and acid indigestion over the years. Some voluntarily took their products off the market as soon as the NDMA finding was reported by the FDA.

Pharmaceutical Technology
JUNE 7, 2023
The US Food and Drug Administration (FDA) has accepted GSK’s supplemental biologics licence application (sBLA) for its Jemperli (dostarlimab) plus chemotherapy for priority review. The regulatory body aims to take a decision on the application by 23 September 2023 under the Prescription Drug User Fee Act.
Pharmaceutical Technology
MAY 31, 2023
The US Food and Drug Administration (FDA) has accepted Bristol Myers Squibb’s new drug application (NDA) for repotrectinib for priority review. The FDA has assigned a prescription drug user fee act action date of 27 November 2023 for the NDA. Its favourable properties for penetrating the human brain increase intracranial activity. “We

Celeritas
MARCH 3, 2023
Predictive : future trends based on past trends Prescriptive : what to do based on the current situation and future likelihoods. A highly accurate way to prescribe drugs could be made possible by prescriptive analytics. These threaten our marine ecology as well as our future source of food.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content