Food Insecurity is a Leading Health-Related Social Need Among Patients With Diabetes
Pharmacy Times
MAY 2, 2023
Screening tools are becoming widely used to help clinicians address the needs of patients with diabetes to improve health outcomes.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmacy Times
MAY 2, 2023
Screening tools are becoming widely used to help clinicians address the needs of patients with diabetes to improve health outcomes.
Pharmaceutical Technology
FEBRUARY 23, 2023
Lactobacilli-based probiotics is a key innovation area in environmental sustainability Probiotics are live bacteria and yeasts, found in yogurt and other food supplements that provide health benefits when ingested or applied to the body. Nestle is the leading patent filer of lactobacilli-based probiotics.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

European Pharmaceutical Review
MARCH 8, 2023
Now working as a woman in pharma, Dr Ekaterina Malievskaia highlighted her female inspirations in the industry, two of which have held leading roles at Bristol Myers Squibb and the US Food and Drug Administration (FDA). Click here to read EPR’s previous ‘ Women Inspiring Pharma’ series.

Pharmaceutical Technology
OCTOBER 11, 2022
Swiss biotech company Stalicla has signed a licensing deal with Evgen Pharma for the latter’s lead asset, SFX-01, in neurodevelopmental disorders and schizophrenia. in milestone payments that include $5m upon receipt of the investigational new drug (IND) from the US Food and Drug Administration (FDA), which is expected late next year.

Spotio
OCTOBER 28, 2022
An effective B2B sales lead generation program will propel your company forward. According to a recent study, 61% of B2B marketers say that lead generation is their top challenge. First, sales and marketing teams need to work together to generate and vet quality leads. Search B2B Lead Databases. What’s the solution?
Pharmaceutical Technology
JULY 12, 2022
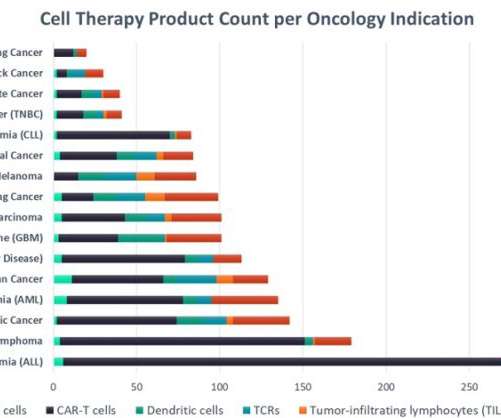
The success of chimeric antigen receptor T-cells (CAR-T) in blood cancers has led to the US Food and Drug Administration's (FDA) approval of six products with a pipeline of cell therapies that numbers in the thousands.
Pharmaceutical Technology
MAY 15, 2023
Innovation S-curve for the pharmaceutical industry Periodontitis drugs is a notable innovation area in the pharmaceutical industry Periodontal diseases, which may lead to the damage of teeth or bone, are caused by infections and inflammation of the gums and bone that surround and support the teeth.
Pharmaceutical Technology
FEBRUARY 14, 2023
It helps to break down food, absorb nutrients and fight foreign micro-organisms that cause diseases. Novo Nordisk Foundation is one of the leading patent filers for lactobacilli-based drug compositions. The company has several lactobacillus-based probiotic products, infant formulas, yogurts, and drinks.

World of DTC Marketing
JUNE 4, 2021
Mike always battled weight issues, but since he went out on disability, he had gained more weight, leading to diabetes. An unhealthy diet is one of the leading risk factors for poor health and accounts for up to 45% of all deaths from these cardiometabolic diseases, the researchers noted. 3hree: Healthier foods can cost more.

World of DTC Marketing
AUGUST 22, 2022
Globally, nearly half of deaths due to cancer can be attributable to preventable risk factors, including three leading risks of smoking, drinking too much alcohol, or having a high body mass index, a new paper suggests. 3: Obesity is caused by poor access to healthy food, primarily fruits and vegetables.

World of DTC Marketing
AUGUST 31, 2022
“Obesity is a leading cause of preventable disease and death among U.S. Foods lacking in the Western diet-whole grains, vegetables, fruits, and nuts-seem to help with weight control and also help prevent chronic disease. and see the gigantic platters of food in front of them. at the National Institutes of Health.

Pharmacy Times
JANUARY 29, 2024
Specific drugs, foods, and supplements can lead to this condition.

World of DTC Marketing
JUNE 6, 2022
Diabetes can lead to serious health complications like heart disease and stroke, so it’s important to take steps to avoid developing the disease if possible. There are many risk factors for type 2 diabetes, but one of the biggest is a poor diet high in processed foods and added sugar.
European Pharmaceutical Review
MARCH 18, 2024
He added that the trial results could lead to this specific infusion pump delivery system to be approved by regulatory bodies such as the US Food and Drug Administration (FDA).

European Pharmaceutical Review
MAY 12, 2023
Innovative therapies for rare diseases The acquisition will help to drive the growth of CTI’s lead product, kinase inhibitor VONJO ® (pacritinib) “in treating myeloproliferative disease,” stated Dr Adam Craig, President, Chief Executive Officer and Interim Chief Medical Officer of CTI BioPharma.

World of DTC Marketing
NOVEMBER 1, 2020
Fauci, the country’s leading infectious-disease expert, said in a wide-ranging interview late Friday. Fauci, a leading member of the government’s coronavirus response, said the United States needed to make an “abrupt change” in public health practices and behaviors. We’re in for a whole lot of hurt.

European Pharmaceutical Review
DECEMBER 5, 2023
Incretins are “gut hormones that are secreted after food intake and play a role in modulating blood glucose by stimulating insulin secretion and suppressing appetite,” Roche highlighted. Roche has agreed to acquire biotech Carmot Therapeutics for an upfront purchase price of $2.7

World of DTC Marketing
DECEMBER 2, 2021
” So, are we supposed to believe that better images in DTC ads lead to patients asking for an Rx? Ad recall is essential for CPGs, but for prescription drugs, the question that’s missing is “what action did it lead to? Higher numeracy was associated with better gist and verbatim recall.” ” Uh…no.

Clarivate
MARCH 14, 2024
Over the past few years nitrosamines and nitrosamine impurities have been increasingly gaining the reputation of being high-risk containments in the food industry. The food sector has already initiated the publication of preliminary regulations aimed at restricting PFAS in packaging. [3].

Pharmaceutical Technology
JUNE 21, 2023
AGEPHA Pharma has received approval from the US Food and Drug Administration (FDA) for LODOCO (colchicine, 0.5 We are dedicated to addressing heart disease, the leading cause of death, by ensuring all patients have access to LODOCO.” mg tablet) to treat cardiovascular disease.
Pharmaceutical Technology
JUNE 8, 2023
The US Food Drug Administration (FDA) has granted orphan drug designation to DTx Pharma’s investigational DTx-1252 for the treatment of Charcot-Marie-Tooth disease Type 1A (CMT1A). CMT1A is a progressive neuromuscular autosomal-dominant disease that leads to life-long loss of muscle function, as well as disability.
Pharmaceutical Technology
JUNE 6, 2023
The therapy has also received orphan drug designation and breakthrough therapy designation from the US Food and Drug Administration. Vertex Pharmaceuticals founder and newly arrived executive chair of Alkeus Joshua Boger stated: “Stargardt is a progressive, debilitating disease that leads inevitably to devastating vision loss.

World of DTC Marketing
JUNE 5, 2021
There is disagreement whether obesity is really considered a disease or a behavioral risk factor, similar to smoking, alcohol, and substance abuse that may lead to disease. Some of the environmental conditions that lead to obesity are physical activity, alcohol consumption, socioeconomic status, parent feeding behavior, and diet.

European Pharmaceutical Review
NOVEMBER 27, 2023
This leads to a lipid build-up in vital organs such as the liver, blood vessels and other tissues, and is usually fatal for these individuals within their first twelve months of life, Alexion reported. This ultra-rare metabolic disease occurs due to lysosomal acid lipase (LAL) enzyme deficiency.

World of DTC Marketing
FEBRUARY 10, 2022
30, 2021, the study’s lead author, Thomas J. It isn’t real food; it is the packaged, highly refined, chemically-laden products marketed to us as “food” that is killing us and will continue to do so until we reframe what is being perpetrated on consumers in terms of addiction. . 1, 2020, and Sept.
Pharma Leaders
MARCH 20, 2023
The US Food and Drug Administration (FDA) has granted fast track designation to Arrowhead Pharmaceuticals’ ARO-APOC3 that helps to lower triglycerides in adult patients with familial chylomicronemia syndrome (FCS). Earlier, ARO-APOC3 received Orphan Drug designation from the FDA and the European Union.

European Pharmaceutical Review
APRIL 17, 2024
Roche multiple sclerosis subcutaneous injection: late-breaking data “Updated results from OCARINA II further underline the potential benefits of subcutaneous OCREVUS for patients with both relapsing and progressive forms of MS,” shared Scott Newsome , DO, lead author, Johns Hopkins University School of Medicine.

World of DTC Marketing
FEBRUARY 1, 2022
According to STAT News “after a double-digit decline in 2021, the sector has fallen another 20% in the new year, erasing billions in value and leading even the most seasoned investors to question whether biotech has further to fall. Meanwhile, the Food and Drug Administration has delayed several drug approvals, and Sen.

European Pharmaceutical Review
NOVEMBER 28, 2023
The US Food and Drug Administration (FDA) has approved the first drug for desmoid tumours (desmoid fibromatosis), an oral gamma secretase inhibitor. SpringWorks Therapeutics’ Ogsiveo (nirogacestat) tablets are authorised for adults with progressing desmoid tumours who require systemic treatment.
Pharmaceutical Technology
AUGUST 23, 2022
The US Food and Drug Administration (FDA) has granted Orphan Drug Designation to Ocelot Bio’s OCE-205 to treat hepatorenal syndrome. A lead candidate of the company, OCE-205 is a peptide therapeutic. Currently, there exist no therapies approved by the Food and Drug Administration for treating HRS-AKI.

Pharmaceutical Technology
APRIL 18, 2023
IntelGenx Corp announced that the US Food and Drug Administration (FDA) has approved the company’s Rizafilm VersaFilm new drug application (NDA) for the treatment of acute migraine. The drug constricts blood vessels leading to a reduction in swelling and inflammation of blood vessels associated with migraine.

European Pharmaceutical Review
FEBRUARY 2, 2024
For example, LEQEMBI was the first anti-amyloid beta treatment to be granted traditional US Food and Drug Administration (FDA) approval. Eisai is leading development of LEQEMBI and regulatory submissions globally. Eisai and Biogen are co-commercialising and co-promoting the treatment.
Pharmaceutical Technology
OCTOBER 7, 2022
Provention Bio and Sanofi US have signed a co-promotion agreement to launch the former’s lead investigational drug candidate, teplizumab for the delay in the onset of clinical type 1 diabetes (T1D). The US Food and Drug Administration (FDA) is presently reviewing teplizumab for the delay of clinical T1D in people who are at risk.

European Pharmaceutical Review
DECEMBER 22, 2023
The deal includes Karuna’s lead asset KarXT (xanomeline-trospium), a potential first-in-class treatment for schizophrenia. Currently under review by the US Food and Drug Administration (FDA), KarXT is expected to launch in the US in late 2024. per share in cash.
European Pharmaceutical Review
DECEMBER 5, 2023
Currently, imetelstat is being reviewed by the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) for approval in transfusion dependent anaemia in patients with lower risk MDS who have failed to respond or have lost response to or are ineligible for ESAs, according to Geron.

Pharmaceutical Technology
JUNE 19, 2023
UK-based pharmaceutical giant GSK has announced that the US Food and Drug Administration (FDA) has extended the review period of its new drug application (NDA) for the rare bone cancer drug momelotinib by three months. The reason for this review extension was undisclosed in the announcement. people per every 100,000 individuals in the US.
Pharmaceutical Technology
APRIL 3, 2023
With eight marketed biosimilars, Sandoz is offering the broadest biosimilar portfolio and is the leading biosimilars company in Europe, with more than two decades of experience. The citrate-free HCF of Hyrimoz (adalimumab-adaz) injection has also recently received approval from the US Food and Drug Administration (FDA).

Pharmaceutical Technology
APRIL 6, 2023
Leading with Nefecon as our anchor product, we are building a pipeline of promising renal drug candidates to address huge unmet medical needs.” In February 2023, Nefecon received a global innovative product on fast track (GIFT) designation from South Korea’s Ministry of Food and Drug Safety (MFDS).

World of DTC Marketing
FEBRUARY 16, 2022
Pfizer has made billions of dollars from their COVID vaccine but shouldn’t they have a responsibility to warn people that obesity is a leading cause of severe COVID? Treating obesity is not as easy as telling people to exercise more and eat less high-fat foods, but we can’t continue the current status quo.
European Pharmaceutical Review
SEPTEMBER 8, 2023
Tirzepatide has been shown… [to lead] to substantial weight loss of, on average, 15 percent over 70 weeks. The US Food and Drug Administration (FDA) first approved tirzepatide for type 2 diabetes in May 2022. Final guidance is scheduled to be published on 11 October 2023.
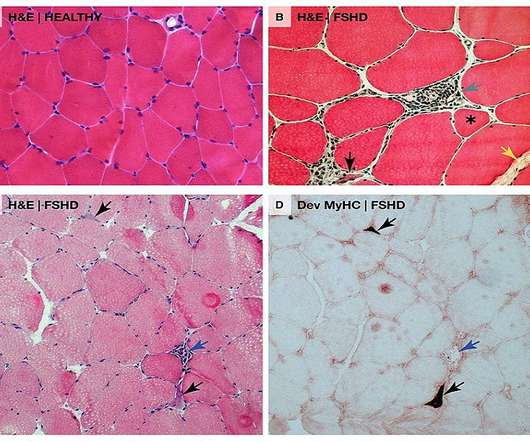
Pharmaceutical Technology
JANUARY 19, 2023
The US Food and Drug Administration (FDA) has granted Fast Track designation for Avidity Biosciences’ AOC 1020 to treat facioscapulohumeral muscular dystrophy (FSHD). This DUX4 protein abnormal expression leads to modifications in gene expression in muscle cells which are associated with progressive muscle function loss in FSHD patients.

European Pharmaceutical Review
JULY 28, 2023
Publication of results of the Phase III SKYLARK study “is a pinnacle moment in treating postpartum depression ( PDD ),” stated Dr Kristina M Deligiannidis, Professor at the Institute of Behavioral Science at the Feinstein Institutes, the trial’s principal investigator, lead author of the paper.

Clarivate
MARCH 17, 2022
While we’re pleased to celebrate these scientists leading the way in sustainability research, these spotlights are not meant to be comprehensive lists of all Highly Cited Researchers contributing to a given SDG; there are many other Highly Cited Researchers that have also contributed to these SDGs, not named here. SDG 1: No Poverty.
European Pharmaceutical Review
AUGUST 11, 2023
This review started after results of a Phase III study (STAND) did not show a statistically significant difference between crizanlizumab and placebo in rates of pain crises leading to a healthcare visit over the first-year. Crizanlizumab is approved for use by the United States Food and Drug Administration (FDA).
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content