AstraZeneca withdraws US COVID vaccine application, shifts focus to antibody treatments
Fierce Pharma
NOVEMBER 10, 2022
AstraZeneca withdraws US COVID vaccine application, shifts focus to antibody treatments. aliu. Thu, 11/10/2022 - 09:02.

Fierce Pharma
NOVEMBER 10, 2022
AstraZeneca withdraws US COVID vaccine application, shifts focus to antibody treatments. aliu. Thu, 11/10/2022 - 09:02.
Pharmaceutical Technology
NOVEMBER 25, 2022
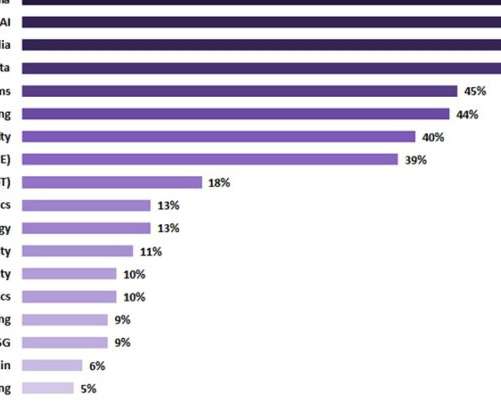
Artificial intelligence (AI) was seen as one of the top current investment priorities and was thought to continue to attract investment in the healthcare sector in the upcoming two years, according to GlobalData's latest report ‘Digital Transformation and Emerging Technology in the Healthcare Industry - 2022 Edition’. In this survey-based report tracker, digital media was prioritised as a top current investment target, with 53% of surveyed respondents confirming that their companies are currentl
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Medgadget
DECEMBER 7, 2022
At the University of South Australia, researchers designed a system that allows them to measure a patient’s blood pressure with a camera. The camera visualizes the patient’s forehead and focuses on two regions in particular to optically determine photoplethysmographic signals that AI algorithms then convert to blood pressure data. The researchers tested their system in 25 volunteers, and so far it has proven to be approximately 90% as accurate as traditional pressure cuff measurements.

MedCity News
OCTOBER 16, 2022
As society becomes more open to and reliant on these tools, it is the responsibility of technology companies, especially those tasked with assisting people’s mental health, to build and maintain artificial intelligence and machine learning models that adapt alongside societal needs.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
pharmaphorum
DECEMBER 19, 2022
A collaboration between academic centres in the UK has won government funding for a digital approach to dysfunctional breathing or dyspnoea, a symptom that affects around 10% of the population. The Engineering and Physical Sciences Research Council (EPSRC) has set aside £400,000 (almost $490,000) for the project, which will help fund work at the Universities of Plymouth, Salford and Stirling, and the Glasgow School of Art.

Clarivate
JULY 26, 2022
Starting from the 2023 JCR release, Journal Impact Factors will be expanded to all Web of Science Core Collection journals including arts and humanities. Today we’re proud to announce that in the 2023 release of the Journal Citation Reports , all Web of Science Core Collection journals will receive a Journal Impact Factor (JIF). This means expanding the JIF from Science Citation Index Expanded (SCIE) and Social Science Citation Index (SSCI) to include journals from the Arts and Humanities Citati
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

PharmExec
JULY 20, 2022
Thursday, July 21st 2022 at 11am EST, 8am PST, 5pm CEST Join this webinar to hear industry thought leaders discuss how data and experiences serve clinical and commercial needs, explore which platforms and technologies can enable a robust digital health ecosystem, and share recommendations for building a digital health strategy fit for your business.

Copyright Clearance Center
NOVEMBER 7, 2022
November 7, 2022, LEHI, Utah — CloudSource+ , the newest content solution for open access materials from SirsiDynix , has integrated with Get It Now from CCC (Copyright Clearance Center) to provide immediate purchase and delivery of full-text articles to unsubscribed journals. Get It Now is used by hundreds of academic libraries around the world to help expand their virtual collections.

PharmaVoice
JUNE 30, 2022
As ‘the Amazon of DNA,’ Twist CSO, Aaron Sato, discusses the company’s breakthrough approach to DNA production and its expansion into drug development.
Pharma Marketing Network
JULY 27, 2022
In the first two weeks after a cancer diagnosis, there are likely dozens of doctor’s visits, tests, consultations, and decision points. The patient care team supplies an abundance of information, although sifting through brochures and online resources can be daunting for the patient and their caregiver. Adhering to regulatory guidelines is an important step pharma takes to ensure that promotional medical materials contain balanced information that is accurate and relevant. 1 The marketer’s goal

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Fierce Pharma
DECEMBER 29, 2022
Biogen, FDA's inappropriate Aduhelm coordination detailed in blistering congressional report. aarmstrong. Thu, 12/29/2022 - 14:42.

Pharmaceutical Technology
NOVEMBER 14, 2022
In a field dominated by antibodies and small molecules, two cell-therapy based approaches have come under the spotlight for showing early signs of efficacy in treating lupus. In September, a group from Friedrich Alexander University Erlangen-Nuremberg reported that five patients with lupus achieved remission after an infusion of autologous chimeric antigen receptor (CAR)-T cells led to a deep depletion of B cells.
Medgadget
AUGUST 22, 2022
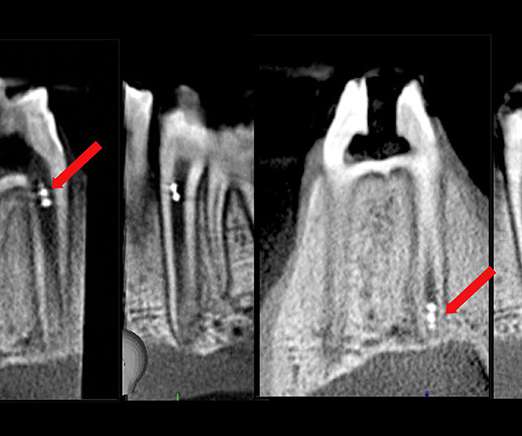
Researchers at the University of Pennsylvania have developed a microrobot system that can help with biofilm disruption, drug delivery, and sample retrieval, all within the restrictive space of the root canal. It can be difficult for dentists to know if they have removed all the infectious material when performing a root canal, and failure to do so will typically result in an infection reoccurring.

MedCity News
OCTOBER 1, 2022
The metaverse holds promising potential to transform healthcare delivery for the better, according to a keynote talk delivered at MedCity’s Invest Digital Health conference. The speaker identified workforce training, patient experience, healthcare facilities and medical manufacturing as the main areas poised for disruption.

Advertiser: ZoomInfo
Marketing technology is essential for B2B marketers to stay competitive in a rapidly changing digital landscape — and with 53% of marketers experiencing legacy technology issues and limitations, they’re researching innovations to expand and refine their technology stacks. To help practitioners keep up with the rapidly evolving martech landscape, this special report will discuss: How practitioners are integrating technologies and systems to encourage information-sharing between departments and pr

pharmaphorum
AUGUST 8, 2022
A study of a mobile health app used to support patients hospitalised with heart failure has concluded that it provided a modest improvement in quality-of-life scores, as well as better clinical outcomes. The finding comes from the large-scale CONNECT-HF cluster study, which looked at various patient engagement and education interventions to see if they could help patients with heart failure do better after discharge from hospital.

Clarivate
JUNE 28, 2022
Today we release the 2022 update to the annual Journal Citation Reports (JCR). This year’s release demonstrates the enormous impact the COVID-19 pandemic has had on scholarly publishing. We also identify and define a new type of anomalous citation behavior: self-stacking. The annual JCR release enables the research community to evaluate the world’s high-quality academic journals using a range of indicators, descriptive data and visualizations.
European Pharmaceutical Review
OCTOBER 13, 2022
Scientists at MIT have demonstrated that a novel robotic capsule could potentially replace conventional biologic injection methods by tunnelling through the intestinal mucus barrier to deliver insulin. The research, published in the journal Science Robotics described how the new drug delivery system can transport large protein and small-molecule drugs, like vancomycin, an antibiotic peptide.

PharmExec
SEPTEMBER 10, 2022
FDA backs the benefits of adopting continuous manufacturing, a strategy which has accelerated approval and launch timelines, and its Emerging Technology Program could be a helpful tool.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

Copyright Clearance Center
DECEMBER 13, 2022
December 13, 20 22 – Danvers, Mass. – CCC , a leader in advancing copyright, accelerating knowledge, and powering innovation, has been named one of the Top Places to Work in Massachusetts in the 15 th annual employee-based survey project from The Boston Globe. CCC ranked #17, up from #33 last year, in the “ large companies ” category (organizations between 250-999 employees).

PharmaTech
OCTOBER 3, 2022
Biopharma focuses on streamlining biomanufacturing and supply chain issues to drive uptake of cell and gene therapies.

PharmaVoice
SEPTEMBER 6, 2022
From standout executives to R&D pioneers — this year’s class of honorees is defining the next era of leadership.

Fierce Pharma
DECEMBER 22, 2022
Pfizer, Sanofi settle first California Zantac case slated for trial: report. zbecker. Thu, 12/22/2022 - 12:38.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Pharmaceutical Technology
AUGUST 4, 2022
Soaring patient numbers are stretching hospital capacity across the globe, forcing healthcare providers and their partners to think laterally about how to meet the demand without doubling their resources. Smarter diagnostic tools and more sophisticated remote- and self-care models will have an increasingly important role to play, as long as any advances are seen to deliver excellent outcomes and a better patient experience.

Medgadget
NOVEMBER 11, 2022
Researchers at the University of Texas at Arlington, in collaboration with Shani Biotechnologies, a local firm, have created a point-of-care device that can accurately measure hemoglobin levels and perform pulse oximetry in individuals with dark skin. At present, methods to determine hemoglobin levels at the point of care, such as pulse oximetry, are inaccurate in individuals with higher levels of melanin in their skin, and there is a clear need to develop alternatives that work for everyone.

MedCity News
NOVEMBER 1, 2022
Large employers, particularly self-insured companies, can demand a better and fairer system. They can use their contracting power to insist that their plans cover all FDA approved drugs.
pharmaphorum
OCTOBER 6, 2022
An artificial intelligence-powered digital health worker has been unveiled by the World Health Organisation (WHO) as its latest tool for disseminating reliable health information to the public. Originally developed by New Zealand tech company Soul Machines with support of the Qatar Ministry of Health, the first version of the virtual health worker was used to combat misinformation about the pandemic.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

Clarivate
JUNE 21, 2022
Each year, the Journal Citation Reports (JCR) release provides a summary of the network of scholarly citations from the prior year of Web of Science coverage. It is a snapshot of the continually evolving research landscape. “It’s tough to make predictions, especially about the future.”. Baseball player Yogi Berra. As we approach this year’s JCR release at the end of this month, we reviewed factors that may affect the published metrics.

European Pharmaceutical Review
JUNE 24, 2022
The Greek state has filed a lawsuit against Novartis , asking for € 214 million in compensation for non-pecuniary damages* the country may have suffered due to alleged illegal payments to government officials and healthcare providers. Commenting on the lawsuit , Greece’s Health Minister Thanos Plevris, stated that the country is seeking compensation for the damage it has suffered “from the actions that Novartis itself has admitted to in the USA that concerns payments to doctors.”.

PharmExec
NOVEMBER 8, 2022
Why enabling a shared culture of quality across an organization is key to maintaining pharma’s recent pace of innovation wins—while inspiring new levels of confidence.

PharmaTimes
OCTOBER 4, 2022
Treatment involves adult patients with severe alopecia areata

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content