Endogena finalises enrolment for retinitis pigmentosa trial
PharmaTimes
MAY 4, 2023
The treatment is a small molecule that activates endogenous retinal stem and progenitor cells

PharmaTimes
MAY 4, 2023
The treatment is a small molecule that activates endogenous retinal stem and progenitor cells

MedCity News
APRIL 30, 2023
A new study found that ChatGPT might actually be quite successful in providing high-quality answers to patient questions during an era in which doctors and nurses are too busy to do so. The research evaluated two sets of answers to patient inquiries — one written by physicians, the other by ChatGPT. A panel of healthcare professionals determined that ChatGPT’s answers were significantly more detailed and empathetic.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PharmaVoice
MAY 1, 2023
The CEO of Ventoux Biosciences has the disease that his company is aiming to treat, and it's driving him to find better medications.

Fierce Pharma
MAY 3, 2023
ImmunoGen touts 'home run' as ovarian cancer drug Elahere extends lives in landmark trial win aliu Wed, 05/03/2023 - 14:50

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Dominic Tyer
MAY 5, 2023
Pharma has an opportunity to make more effective - and wider - use of social media, but companies are not currently meeting HCP expectations

MedCity News
MAY 3, 2023
The Drug Enforcement Administration received 38,000 comments on its proposed rule that would roll back Covid-19 flexibilities for the prescribing of controlled substances via telemedicine. In response, the DEA chose to temporarily extend the flexibilities, a decision telehealth companies like Bicycle Health and Mindbloom are in support of.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.
Fierce Pharma
MAY 2, 2023
Pfizer pulls off Q1 surprise with strong sales even as COVID vaccine demand plummets kdunleavy Tue, 05/02/2023 - 09:27

Pharmaceutical Technology
MAY 1, 2023
Astellas Pharma has entered into a definitive agreement to buy US-based biopharmaceutical company Iveric bio, in a deal valued at nearly $5.9bn. Under the deal terms, the company, through Astellas US Holding’s wholly owned subsidiary Berry Merger Sub, will acquire all the outstanding Iveric Bio shares for $40.00 in cash for each share. Both the companies’ Boards of Directors have unanimously approved the deal.
MedCity News
MAY 3, 2023
In an interview, Rob Bressler, general manager for Teladoc Health’s virtual primary care solution Primary360, talked about the company’s shift from telehealth for urgent care to whole person care.

Medgadget
MAY 1, 2023
Researchers at Northwestern University and the University of Texas at Austin have developed a thin and flexible graphene “tattoo” that can be affixed to the outside of the heart, and which can monitor heart rhythms and apply corrective electrical impulses to treat arrhythmias. The patch is optically clear, allowing the researchers to also use light to monitor and stimulate the heart.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Fierce Pharma
MAY 2, 2023
GSK staffers kick off series of strikes in UK for the first time in drugmaker's history aliu Tue, 05/02/2023 - 14:22

Pharmaceutical Technology
MAY 4, 2023
Forge Biologics and global life sciences company Labcorp have announced a strategic adeno-associated virus (AAV) gene therapy development and manufacturing collaboration. Gene therapy clients will gain access to coordinated scientific expertise, drug development services and AAV manufacturing capabilities, advancing the development of new gene therapies.
MedCity News
MAY 4, 2023
Oncology Ventures recently announced investments in three cancer startups — VivorCare, Gabbi and Health Universe. The fund’s founding partner said each company demonstrated an ability to tackle a specific problem within the cancer care space, such as enabling earlier breast cancer diagnoses and providing personalized care plans for cancer survivors.
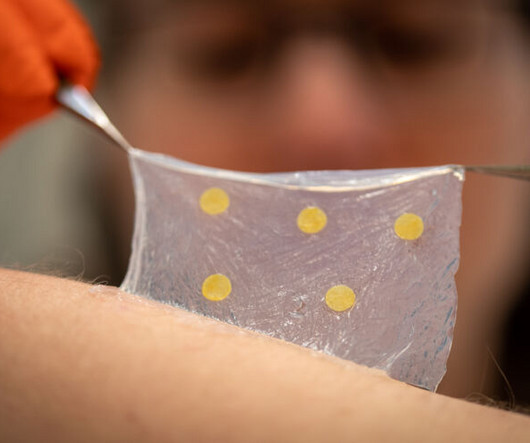
Medgadget
MAY 1, 2023
Researchers at Linköping University in Sweden created a wound dressing that can provide a visual indication if the underlying wound is infected. Chronic wounds are difficult to manage, and presently healthcare staff must remove the dressing regularly to check if an infection is developing. However, this can disrupt the wound by damaging the scab, impairing healing, and may even introduce pathogens in the process.

Advertiser: ZoomInfo
Marketing technology is essential for B2B marketers to stay competitive in a rapidly changing digital landscape — and with 53% of marketers experiencing legacy technology issues and limitations, they’re researching innovations to expand and refine their technology stacks. To help practitioners keep up with the rapidly evolving martech landscape, this special report will discuss: How practitioners are integrating technologies and systems to encourage information-sharing between departments and pr

Fierce Pharma
MAY 4, 2023
Moderna warns of severe sales slowdown in Q2, takes $378M charge amid lull in vaccinations aliu Thu, 05/04/2023 - 10:02

PharmaVoice
MAY 2, 2023
What’s so different about doing pharma business in the U.K. versus Japan? One of Moderna’s top brass shares her tips for navigating choppy cultural waters.
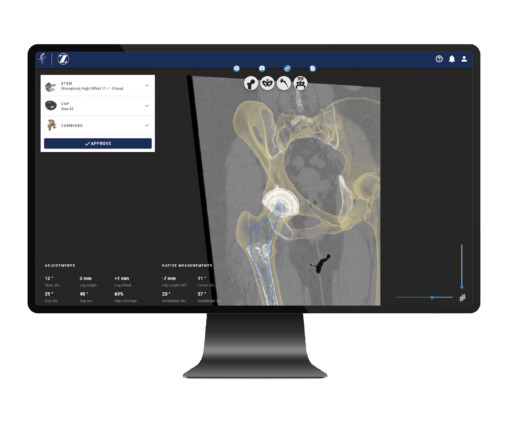
MedCity News
MAY 5, 2023
Formus Labs, a New Zealand-based medical technology startup, has received 510(k) clearance from the FDA for Formus Hip, its fully automated 3D software for hip replacement preoperative planning. The company hopes to start piloting the product at healthcare sites across the U.S. in the next few months.
European Pharmaceutical Review
MAY 2, 2023
Cancer remains a global threat with around 10 million people dying from the disease each year worldwide, making it the second leading cause of death. 1,2 In the EU, an estimated 2.7 million people are diagnosed with cancer annually and the number is growing. 3 With greater scientific understanding of cancer, the industry has made significant progress in developing innovative medicines to help patients.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

Fierce Pharma
MAY 4, 2023
J&J's consumer group Kenvue set for $41B IPO, the largest US market debut in more than a year esagonowsky Thu, 05/04/2023 - 09:58

Legacy MEDSearch
MAY 3, 2023
ORLANDO, Fla., May 3, 2023 – Legacy MEDSearch has been named to the Forbes Best Executive Recruitment Firms and Forbes Best Professional Recruiting Firms out of over 25,000 recruitment and staffing firms in the United States, its fifth consecutive mention since 2019. Forbes Best Recruiting Firms lists are based on votes by industry professionals, who have directly worked with recruiting firms and rankings are earned based on merit.

MedCity News
MAY 5, 2023
Absent the ability to bolster their ranks with more clinicians, what rural health centers need first and foremost to shift to value are technologies that amplify their clinical capacity without further burdening and burning out their core staff.

Pharmaceutical Technology
MAY 4, 2023
Vertex Pharmaceuticals has received approval from the US Food and Drug Administration (FDA) for KALYDECO (ivacaftor) to treat children with cystic fibrosis (CF) aged between one and four months. The approval allows KALYDECO to be used in infants who have at least one mutation in their cystic fibrosis transmembrane conductance regulator (CFTR) gene that is responsive to the therapy, on the basis of clinical and/or in vitro assay results.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
Fierce Pharma
MAY 2, 2023
Poland takes its COVID-19 vaccine fight to Pfizer's shareholders, urging the company to be a good corporate citizen zbecker Tue, 05/02/2023 - 16:34
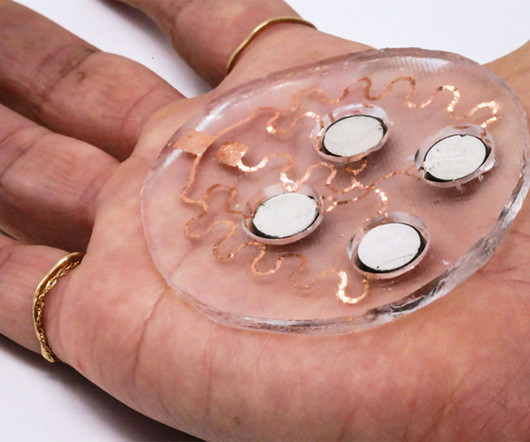
Medgadget
MAY 2, 2023
Engineers at MIT have developed a patch that uses ultrasound to deliver drugs through the skin. Ultrasound can create temporary openings in skin, allowing drugs to pass through. The skin is typically a difficult area for drug delivery, as it forms a tough barrier. However, the patch, which contains piezoelectric transducers, can push drugs through the skin highly efficiently.

MedCity News
MAY 4, 2023
A recent Urban Institute report found that the uninsurance rate for nonelderly people who aren’t citizens will be 39.2% in 2024, about four times higher than it is for the entire U.S. population at 9.8%.
Pharmaceutical Technology
MAY 2, 2023
The US Food and Drug Administration (FDA) has accepted the supplemental biologics licence application submitted by Bristol Myers Squibb for Reblozyl (luspatercept-aamt) as a first-line treatment of anaemia in adults with lower-risk myelodysplastic syndromes (MDS). The regulatory body has also granted priority review to the application and set 28 August 2023 as a target date for the prescription drug user fee act (PDUFA).

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

Fierce Pharma
MAY 1, 2023
Teva, MedinCell win FDA approval for slow-release schizophrenia drug Uzedy kdunleavy Mon, 05/01/2023 - 07:35
PharmExec
MAY 1, 2023
Biogen Inc. announced the approval, a groundbreaking treatment for adults with ALS, offering hope to patients suffering from SOD1-ALS, a rare and fatal genetic form of the disease.

MedCity News
MAY 4, 2023
Resident physicians at Mount Sinai’s Elmhurst Hospital in Queens have voted to authorize a strike. The union that represents these residents has noticed significant disparities in Mount Sinai’s compensation and benefits when compared to the resident physicians employed at the health system’s flagship hospital in Manhattan.
Pharmaceutical Technology
MAY 5, 2023
Sanofi Canada has received approval from Health Canada for its Dupixent (dupilumab injection) to treat eosinophilic esophagitis (EoE) in adults and in adolescent patients aged 12 years and above. It is being jointly developed by Sanofi and Regeneron under a global collaboration agreement. In Canada, it is commercialised by Sanofi Canada and Regeneron Canada.

Advertiser: ZoomInfo
In times of economic uncertainty, account-based strategies are essential. According to several business analysts and practitioners, ABM is a necessity for creating more predictable revenue. Research shows that nearly three-quarters of marketers (74%) already have the resources needed to build successful ABM programs.
Let's personalize your content