‘The Last of Us’ might be fiction, but fungi are a looming threat
PharmaVoice
MARCH 20, 2023
Fungal infections are a growing challenge for hospitalized patients, and new treatments are badly needed.

PharmaVoice
MARCH 20, 2023
Fungal infections are a growing challenge for hospitalized patients, and new treatments are badly needed.

Fierce Pharma
MARCH 22, 2023
AbbVie's blockbuster-to-be Parkinson's combo hits a wall as FDA questions delivery pump aliu Wed, 03/22/2023 - 11:17
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Impetus Digital
MARCH 14, 2023
This is a guest article written by Luke Buchanan, CEO of Redi.Health , and does not necessarily reflect the views and values of Impetus Digital. When we think of adherence in the medical world, it’s often simplified to something along the lines of the degree to which a patient correctly follows medical advice. It’s a simple statement with many, many moving parts, especially when we’re talking about patients with multiple chronic conditions.

Clarivate
MARCH 20, 2023
We have the important responsibility of providing our customers with trustworthy intelligence to help them transform the world for the better. The need for high-quality data from rigorously selected sources is becoming ever more important as the scholarly record becomes increasingly polluted. A trusted record of research is essential for the global R&D community to effectively use published research outcomes to elevate ideas that will benefit everyday life.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

European Pharmaceutical Review
MARCH 9, 2023
A pilot study has demonstrated that a nasal version of the drug Foralumab, an anti-CD3 monoclonal antibody suppressed the inflammatory T cell response and decreased lung inflammation in patients with COVID-19. “This is the first nasal monoclonal antibody—other monoclonal antibody treatments were delivered intravenously and are no longer given as treatment because they are not effective against currently circulating viral variants,” explained Dr Howard Weiner, founder and Director of the Brigham

MedCity News
MARCH 26, 2023
In the EU, there is a widespread distrust of American tech companies, according to a recent conference presentation by an international healthcare lawyer. Because of this, the EU has established a number of new laws protecting its citizens’ data privacy and creating frameworks for the secure exchange of information — laws that American health tech companies will have to comply with if they do business in the EU.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

Fierce Pharma
MARCH 1, 2023
Pfizer, Viatris and more must face 'overarching conspiracy' claims in generic price-fixing suit aliu Wed, 03/01/2023 - 10:03

Medgadget
MARCH 17, 2023
Researchers at Linköping University in Sweden have developed a method whereby the body can ‘grow its own’ electrodes. The minimally invasive technique involves injecting a hydrogel that is laden with enzymes into target tissues. The enzymes interact with molecules that are present in the tissue to change the structure of the gel and allow it to become electrically conductive.

PM360
MARCH 24, 2023
Technology evolves fast. Nothing illustrates that better than attending SXSW each year and noticing how much has changed in just 12 months. In 2022, SXSW was all NFTs and metaverse activations and events, and in 2023 they weren’t anywhere to be found. But a lot was shared at this year’s SXSW that had staying power. In place of NFTs, psychedelics were given their own conference track this year.
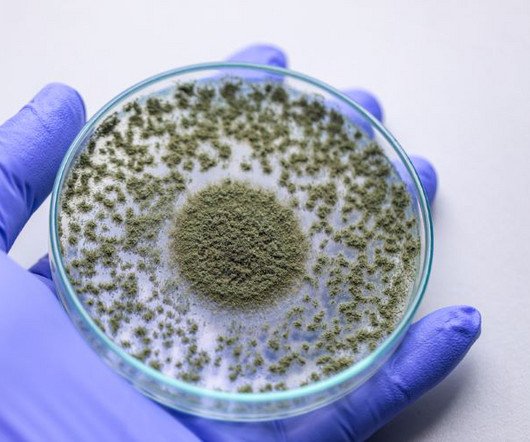
European Pharmaceutical Review
MARCH 22, 2023
In a recent video , pharmaceutical microbiology expert Tim Sandle highlighted areas of concern and attention relating to mycological contamination control in manufacturing. Pharmaceutical product recalls due to fungal contamination have been increasing. These, together with environmental monitoring trend data, highlight several fungal contamination issues associated with pharmaceutical cleanrooms , cold rooms and other controlled areas.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

MedCity News
MARCH 3, 2023
As regulatory processes and reimbursements are refined, there needs to be a continued focus on how to maximize the benefits for patients and ensure DTx can play a role in improving outcomes. The key to this is integration into the clinical workflow.
Pharmaceutical Technology
MARCH 21, 2023
Pre-clinical-stage biotechnology firm InvisiShield Technologies has partnered with Gladstone Institutes to develop intranasal preventatives against airborne viral infections including influenza, respiratory syncytial virus (RSV) and SARS-CoV-2. Under the partnership terms, InvisiShield will offer technical support, as well as funding to develop the intranasal preventatives.

Fierce Pharma
MARCH 10, 2023
Pfizer hopes to nose out competition with approval for new migraine nasal spray Zavzpret kdunleavy Fri, 03/10/2023 - 10:22

Medgadget
MARCH 9, 2023
Researchers at University College London and at the University of Ghana have developed a smartphone-based system that can detect anemia through simple photos taken using the phone’s camera. The technology is intended for use in low- and middle-income countries where access to routine medical diagnostics may be unreliable. The process involves obtaining images of areas of the body which are least pigmented, including the white of the eye, the lower eyelid, and the lip.

Advertiser: ZoomInfo
Marketing technology is essential for B2B marketers to stay competitive in a rapidly changing digital landscape — and with 53% of marketers experiencing legacy technology issues and limitations, they’re researching innovations to expand and refine their technology stacks. To help practitioners keep up with the rapidly evolving martech landscape, this special report will discuss: How practitioners are integrating technologies and systems to encourage information-sharing between departments and pr

PharmExec
MARCH 8, 2023
According to the 2023 State of Revenue Report, pharma companies are prioritizing digital transformation while struggling with inflation and supply chain disruptions.

European Pharmaceutical Review
MARCH 10, 2023
Today marks the opening the NHS Blood and Transplant (NHSBT)’s new Clinical Biotechnology Centre (CBC) in Bristol. The state-of-the-art facility was part-funded by a £9.43 million Government grant and is designed to expand the UK’s ability to manufacture new gene and cell therapies. The CBC will help the UK grow its cell and gene therapy industry in a rapidly growing international market.” “The CBC will help the UK grow its cell and gene therapy industry in a rapidly growing international

MedCity News
MARCH 30, 2023
The recent growth in RPM has demonstrated its importance and potential to reduce or eliminate barriers and improve access to care for patients. By leveraging RPM and other digital health solutions as an important part of a multi-prong approach to care delivery, a future where all patients have equal access to high-quality healthcare becomes a closer reality.

Pharmaceutical Technology
MARCH 14, 2023
The National Health Surveillance Agency (ANVISA) in Brazil has granted approval for Takeda ’s dengue virus vaccine candidate, Qdenga (Dengue Tetravalent Vaccine [Live, Attenuated]) (TAK-003). The vaccine has received approval for preventing dengue disease in people aged four years to 60 years. Qdenga has been developed based on a live-attenuated dengue serotype 2 virus that offers the genetic backbone for four dengue virus serotypes and is designed to provide protection against any of these.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

Fierce Pharma
MARCH 30, 2023
As deadly pathogen spreads, GSK throws a lifeline to Scynexis with $90M deal to market antifungal aliu Thu, 03/30/2023 - 10:39
Medgadget
MARCH 27, 2023
Researchers at the Georgia Institute of Technology have developed a nanotechnological solution for lymphedema, a failure of the lymphatic system that results in uncomfortable and irreversible fluid retention. Previous research efforts have focused on trying to grow new lymphatic vessels, but these researchers have taken a different approach, and instead engineered a drug delivery technology that can directly target sluggish lymphatic vessels and kickstart their pumping action.

PharmExec
MARCH 7, 2023
Latest investigation signals more scrutiny of pharmacy benefit managers and their role in treatment cost and coverage.

European Pharmaceutical Review
MARCH 14, 2023
Pfizer has announced it will acquire Seagen Inc. for a total enterprise value of $43 billion. “Together, Pfizer and Seagen seek to accelerate the next generation of cancer breakthroughs and bring new solutions to patients by combining Seagen’s antibody -drug conjugate (ADC) technology… with Pfizer’s capabilities and expertise,” shared Dr Albert Bourla, Pfizer Chairman and Chief Executive Officer.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

MedCity News
MARCH 2, 2023
Despite ketamine-assisted therapy being approved for treating several mental health conditions, it’s generally not covered by insurance or employers. Though that likely won’t always be the case, as psychedelics gather more research and employers notice the benefits for their workplaces.
Pharmaceutical Technology
MARCH 31, 2023
The European Medicines Agency (EMA) has recommended HIPRA’s Covid-19 vaccine , Bimervax, as a Covid-19 booster. Available to people ages 16 years and above who have been vaccinated with a Covid-19 mRNA vaccine, EMA’s Human Medicines Committee concluded the vaccine is ready for marketing authorization in the EU, on 30 March. Bimervax is a recombinant protein subunit vaccine, marketed by the Girona, Spain-based Hipra.

Fierce Pharma
MARCH 1, 2023
Eli Lilly slashes insulin prices by 70%, caps out-of-pocket costs in major access overhaul fkansteiner Wed, 03/01/2023 - 08:40

PharmaVoice
MARCH 28, 2023
The Supreme Court will now decide whether Amgen can protect Repatha as a whole class of drugs or whether Sanofi’s rival product Praluent can retain a place in the market.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

PM360
MARCH 24, 2023
Cancer-support programs provide vital assistance to patients and their caregivers as they face one of the toughest times of their lives. Yet while patients are interested in these programs, new survey data suggests they aren’t taking advantage of them. Results from 825 patients diagnosed with or treated for cancer who were surveyed on Phreesia’s PatientInsights platform as they checked in for their doctors’ appointments revealed that few cancer patients are accessing the support resources availa

European Pharmaceutical Review
MARCH 27, 2023
A report has predicted the rapid microbiology testing market size is estimated to grow at a compound annual growth rate (CAGR) of 9.31 percent between 2022 and 2027. According to the data, the market is forecasted to increase by $2,487.91 million. The research includes historic market data from 2017 to 2021, using 2022 as the base year. It covers various testing methods such as growth-based, nucleic-acid-based and viability-based.

MedCity News
MARCH 26, 2023
To build remote patient monitoring technology for ready adoption, repetitive data streams must be avoided. To be successful in clinical adoption, we offer technology companies the following three recommendations.

Pharmaceutical Technology
MARCH 28, 2023
Following on from its Covid-19 vaccine programmes, BioNTech has set its sights on a range of infectious diseases for vaccine development. In its FY 2022 report, BioNTech has identified herpes simplex virus (HSV), malaria, and shingles as disease targets. The company saw major successes with its Covid-19 vaccine, developed in collaboration with Pfizer.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content