The ascent of pharma marketing
Dominic Tyer
SEPTEMBER 30, 2022
Pharmaceutical companies with a chief marketing officer are thin on the ground, but their number will shortly see a slight increase thanks to a new appointment at Pfizer

Dominic Tyer
SEPTEMBER 30, 2022
Pharmaceutical companies with a chief marketing officer are thin on the ground, but their number will shortly see a slight increase thanks to a new appointment at Pfizer
European Pharmaceutical Review
SEPTEMBER 30, 2022
Professor Hongsoo Choi’s team at The Department of Robotics and Mechatronics Engineering at the Daegu Gyeongbuk Institute of Science & Technology (DGIST), Korea, developed revolutionary technology that produces over 100 microrobots per minute. The collaboration with Professor Sung-Won Kim’s team at Seoul St. Mary’s Hospital, Catholic University of Korea and Professor Bradley J.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
SEPTEMBER 30, 2022
After several setbacks, bluebird bio bounces back with two major FDA gene therapy approvals. Last month, Zynteglo (betibeglogene autotemcel), or beti-cel, was approved as a one-time potentially curative gene therapy for patients with beta-thalassaemia who require regular blood transfusions. Shortly after this, the FDA announced the accelerated approval of bluebird's Skysona (elivaldogene autotemcel), or eli-cel, on 19 September.

MedCity News
SEPTEMBER 30, 2022
Virtual specialty care networks may provide the sort of longitudinal healthcare relationship we haven’t seen since the era of house calls and doctors’ bags—benefiting patients and physicians alike.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Medico Reach
SEPTEMBER 30, 2022
Teledentistry has made dental services easily accessible to patients. Customers can avail dental services right from their comfort. It includes online consultation via video calls, text messages, or live chat. That’s why the teledentistry industry is gradually evolving. In fact, the industry will likely cross $2105.76 million by 2028 , with more teledentistry companies entering the market.

MedCity News
SEPTEMBER 30, 2022
There are a few key priorities providers and digital health companies should consider in their efforts to better reach patients — including scaling up their commitment to value-based care and working to engage patients beyond their episodic care needs — according to a recent healthcare innovation panel.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

MedCity News
SEPTEMBER 30, 2022
When patients can travel to a pharmacy or doctor’s office in their own neighborhood, they can reach the site on foot or using public transit. They also have to take less time off work and request childcare less often.

Spotio
SEPTEMBER 30, 2022
Whether you’re a software entrepreneur, an executive, or a sales manager , one of the best things that you can do to set yourself up for success is to write a sales action plan that supports your company and helps it to grow. Unfortunately, a lot of business leaders struggle with developing a strategy that can enrich and empower their operations. A sales action plan is the easiest way to lay out your objectives, strategies, and tactics for success, in a way that your whole team can understand.

MedCity News
SEPTEMBER 30, 2022
Novo Nordisk is acquiring global rights to a Ventus Therapeutics drug candidate with potential applications in cardiometabolic disorders such as NASH and chronic kidney disease. The small molecule blocks a protein that has become an attractive target for big pharmaceutical companies, such as Roche and Novartis.
pharmaphorum
SEPTEMBER 30, 2022
Patient organisations have been celebrating the FDA approval yesterday of Amylyx’ amyotrophic lateral sclerosis (ALS) therapy Relyvrio, after not one but two advisory committee meetings that arrived at different conclusions about the drug. The approval makes Relyvrio (sodium phenylbutyrate and taurursodiol) the first new therapy option for ALS – also known as motor neuron disease (MND) – since Mitsubishi Tanabe Pharma’s intravenous Radicava (edaravone) was cleared by the FDA in 2017.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

MedCity News
SEPTEMBER 30, 2022
Employers are having a difficult time navigating all the point solutions in healthcare. But it’s not necessarily a bad thing, as its generating more competition and forcing companies to get creative. Several experts discussed the topic at MedCity’s Invest Digital Health conference.

pharmaphorum
SEPTEMBER 30, 2022
Part I. Novel oncologic drugs continue to command premium pricing, driven by a combination of unmet needs, and delivering often-iterative, sometimes-significant improvements in survival or maintenance of a progression-free state. The stable of PD-1/PD-L1 checkpoint inhibitors, led by Keytruda (pembrolizumab), represents the current generation of high value, high price drugs to tackle a variety of cancers in mono- or combination therapy.

MedCity News
SEPTEMBER 30, 2022
While EHRs are often viewed as essential clinical tools, most were originally designed to facilitate billing operations. By incorporating clinically responsive workflows that readily present the right information to clinicians at the point of care, EHRs can finally fulfill their destiny – to drive better patient care.

Copyright Clearance Center
SEPTEMBER 30, 2022
September 29, 2022 – Danvers, Mass. – CCC , a leader in advancing copyright, accelerating knowledge, and powering innovation, announced today that RightFind Cite It is now available for Mac users. RightFind Cite It is CCC’s easy-to-use reference management tool that automatically formats bibliographies directly within Microsoft Word with the style and citations its customers use, saving manual work and time.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

MedCity News
SEPTEMBER 30, 2022
Of all systematic changes to healthcare, these four adjustments would be easy enough to roll out quickly and would have a massive impact on the country’s health outcomes.

Map My Customers
SEPTEMBER 30, 2022
In a world of continual rising costs and inflation, it is more crucial now than ever to implement ways to minimize and control costs within your business. Everyone within an organization can do their part to contribute to this effort including those within the sales team. A sales team is constantly on the go visiting customers and fuel costs on these travels play a huge role in affecting overall operating costs.

MedCity News
SEPTEMBER 30, 2022
An Amylyx Pharmaceuticals drug developed to slow the progression of amyotrophic lateral sclerosis is now approved, making it just the third FDA-approved treatment for the disease. Amylyx will market its new product under the name “Relyvrio.”.

Copyright Clearance Center
SEPTEMBER 30, 2022
The post CCC Launches OA Agreement Intelligence appeared first on Copyright Clearance Center.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

pharmaphorum
SEPTEMBER 30, 2022
Denmark’s Novo Nordisk has agreed to pay $70 million to license rights to a drug developed by Ventus Therapeutics with potential in non-alcoholic steatohepatitis (NASH) and several other diseases associated with inflammation. The deal – which includes additional milestones of up to $633 million plus tiered royalties on sales and R&D funding – focuses on VENT-01, the lead drug in Ventus’ peripherally-restricted NLRP3 inhibitor programme, which is currently in preclinical developme

Integrity Solutions
SEPTEMBER 30, 2022
It is no secret that costs are on the rise and that finding justification for a price increase is as easy as looking at everyday purchases. From higher grocery bills to soaring costs for the materials and resources required to produce and deliver products and services, we’ve all felt the impact of inflation and other factors that have steadily pushed prices up over the past year or so.
pharmaphorum
SEPTEMBER 30, 2022
Merck & Co has been fighting to defend its blockbuster type 2 diabetes therapy Januvia from generic competition for years, and a new judgment looks set to protect its third biggest-selling drug for another few years. The latest verdict – in a federal appeals court – has upheld the validity of a key patent on sitagliptin, the active ingredient in Januvia , as well as metformin combinations Janumet and Janumet XR.

MedTechVets
SEPTEMBER 30, 2022
SEPTEMBER 30 – For the past several months, MedTechVets was fortunate to be joined by Military Transition and Corporate Veteran Readiness Specialist , Sara Blewett. Her expertise was utilized to evaluate MTV’s program effectiveness and implement improvements to ensure a sustainable future to assist veterans to their career transition goals. “Sara is such a wonderful bundle of knowledge!

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Pharmaceutical Technology
SEPTEMBER 30, 2022
InflaRx has submitted an application seeking Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA) for its lead candidate, vilobelimab, to treat Covid-19 patients who are critically ill. The latest development comes after the company had encouraging discussions with the agency at a Type B meeting. In addition, vilobelimab received Fast Track designation from the FDA to treat critically ill, mechanically ventilated, intubated patients with the infection.

CLD, Inc.
SEPTEMBER 30, 2022
The most impactful and dynamic training deliverables in life sciences focus on the patient. These pieces can help learners have and convey empathy for the burden of disease, discuss the limits and possibilities of treatment, perceive more of the hidden consequences of a disease, and learn about patient and caregiver "wish lists" for managing their disease -- ultimately helping learners better position treatments to HCPs.
Pharmaceutical Technology
SEPTEMBER 30, 2022
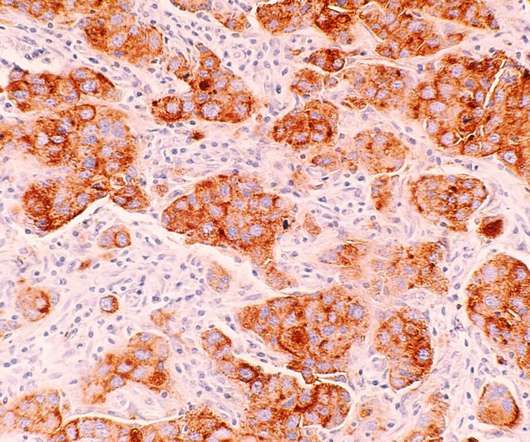
Pheon Therapeutics has announced its launch to progress new antibody-drug conjugates (ADCs) for treating solid tumours. The launch comes after the company raised $68m in a series A financing round led by Brandon Capital, Forbion and Atlas Venture. Seed investor Research Corporation Technologies (RCT) also took part in the funding round. The latest financing will aid the company in progressing its lead ADC programme to clinical proof-of-concept (PoC) and validate the new ADC pipeline. .

PharmExec
SEPTEMBER 30, 2022
In episode 118, Paula Brown Stafford, CEO of Novan, discusses the company’s upcoming NDA filing for its topical agent for molluscum contagiosum, what commercialization could look like and her goals for the future.

Advertisement
Clinical development organizations face a wide array of challenges when it comes to data, many of which can impact the operational effectiveness of their clinical trials. In this whitepaper, experts from Revvity Signals explore how solutions like TIBCO® Spotfire® enable better, more streamlined studies. The whitepaper also features a success story from Ambrx, a leading biopharmaceutical company, detailing how it has leveraged Spotfire to tackle data quality and collaboration challenges in clinic
Pharmaceutical Technology
SEPTEMBER 30, 2022
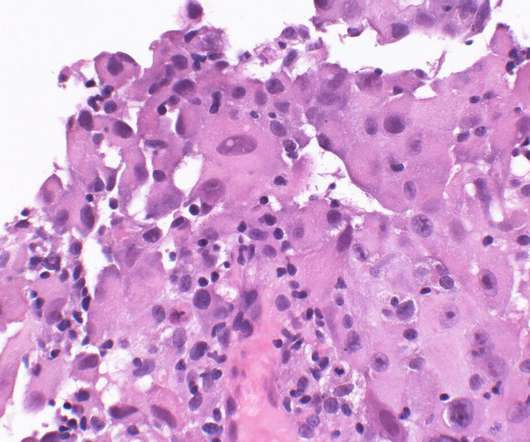
The US Food and Drug Administration (FDA) has granted orphan drug designation (ODD) to Verismo Therapeutics’ SynKIR-110 to treat mesothelin-expressing mesothelioma patients. A next generation cell therapy approach, SynKIR-110 acts on solid tumours. It is said to be the first product to leverage the new KIR-CAR platform, a modified NK-like receptor for boosting efficacy and persistence against aggressive solid tumours.
pharmaphorum
SEPTEMBER 30, 2022
Biogen and Eisai are riding high on the wave of optimism set in motion by the top-line data drop for Alzheimer’s therapy lecanemab – but a number of breakwaters could lie ahead. First and most obviously is the detailed data from the Clarity AD study reported this week, which will be revealed at the Clinical Trials on Alzheimer’s Congress (CTAD) in November.

PharmaTimes
SEPTEMBER 30, 2022
New formulation may be suitable for large-scale trials and commercial supply in the future

pharmaphorum
SEPTEMBER 30, 2022
Trusted Johnson & Johnson brands including Tylenol, Aveeno, Neutrogena, Listerine, and Band-Aid are set to fall under the new, independent company name in a next-chapter move of repurposing and modernisation. Johnson & Johnson’s Kenvue – previously the planned New Consumer Health Company – is just one part of the 135-year old company’s plan to reinvigorate its corporate image.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content