NICE gives thumbs-down to 5 COVID-19 drugs including AZ's Evusheld and Merck's Lagevrio
Fierce Pharma
NOVEMBER 16, 2022
NICE gives thumbs-down to 5 COVID-19 drugs including AZ's Evusheld and Merck's Lagevrio. kdunleavy. Wed, 11/16/2022 - 09:44.

Fierce Pharma
NOVEMBER 16, 2022
NICE gives thumbs-down to 5 COVID-19 drugs including AZ's Evusheld and Merck's Lagevrio. kdunleavy. Wed, 11/16/2022 - 09:44.
MedCity News
NOVEMBER 16, 2022
Risk-based quality management is a data monitoring approach that accelerates the drug development pathway without compromising on safety – and one that is enhanced by the use of technologies such as artificial intelligence.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
NOVEMBER 16, 2022
Novartis targets eye, respiratory units for potential sell-offs after Sandoz: Bloomberg. aliu. Wed, 11/16/2022 - 10:15.

pharmaphorum
NOVEMBER 16, 2022
Novartis has already spun out its eyecare business Alcon, and is now considering the sale of its ophthalmology and respiratory medicines businesses as it continues a narrowing of its focus, according to media reports. A Bloomberg article citing people close to the matter claims that Novartis is engaged in early discussions about the sale of the two units, which could raise billions of dollars in capital that could be reinvested into the pharma group’s pipeline.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Fierce Pharma
NOVEMBER 16, 2022
With Arcutis' steroid-free cream off to the races in plaque psoriasis, company pads case for eczema expansion. fkansteiner. Wed, 11/16/2022 - 09:31.

MedCity News
NOVEMBER 16, 2022
Researchers project that the U.S. healthcare industry will be short 2.1 million nurses by 2025. Including them in reimbursement models might be the most effective way to address nurses’ mass exodus from the profession, according to Rebecca Love, who is a nurse and the chief clinical officer of tech-enabled nurse staffing platform IntelyCare.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

MedCity News
NOVEMBER 16, 2022
Intermountain’s former chief medical officer argued that even though hospital finances are dire, hospitals should prioritize patient experience investments. Her reasoning? People like to talk about their experiences receiving healthcare — both good and bad.
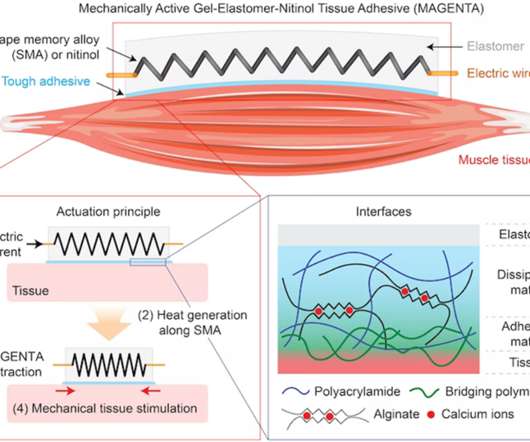
Medgadget
NOVEMBER 16, 2022
Scientists at the Wyss Institute for Biologically Inspired Engineering at Harvard have developed a mechanically active gel-elastomer-nitinol tissue adhesive, otherwise known as MAGENTA. The implantable device functions as a soft robot, and it can be adhered to the outside of a muscle. When an electrical charge is applied to the device, a spring inside made from nitinol (a shape memory alloy) heats up and begins to actuate, creating a contraction and stretching effect on the attached muscle.

MedCity News
NOVEMBER 16, 2022
Because of bad digital experiences, 41% of patients have changed providers, the Notable survey discovered. The findings were presented at the HLTH conference in Las Vegas.

European Pharmaceutical Review
NOVEMBER 16, 2022
As the emphasis of new drug development gravitates towards new, ground-breaking therapies and vaccines, the requirements of manufacturing sites, equipment and processes need to be appropriate for this new environment. For young biotechs, scaling up production brings green-field challenges, while for more established pharma a shift may be required to ensure that facilities are optimised for new ways of working.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

pharmaphorum
NOVEMBER 16, 2022
The 2022 edition of the Access to Medicine Index (ATMI) – which places the top 20 pharma companies for their efforts to improve delivery of medicines to lower income countries – sees GSK retain the number one position, newly joined by Johnson & Johnson in second and AstraZeneca in third. GSK scored highly as its R&D programmes encompass a sizeable chunk of the 83 high burden priority diseases covered in the assessment, an integrated access to medicine strategy in its governance, and robu

MedCity News
NOVEMBER 16, 2022
Historically having worked with Medicaid and Medicare Advantage populations, Uber Health is now expanding its services to self-insured employers. The company made the announcement at the HLTH conference in Las Vegas.

pharmaphorum
NOVEMBER 16, 2022
Last month, pharmaphorum spoke with Dr Charles C. Wykoff about the results from Apellis Pharmaceuticals’ OAKS study on intravitreal pegcetacoplan for geographic atrophy (GA), otherwise known as atrophic or dry age-related macular degeneration (AMD), a leading cause of blindness worldwide. Having presented the data from the 24-month phase 3 trial at the American Academy of Ophthalmology (AAO) annual meeting, Dr Wykoff set aside some time to go into further detail with pharmaphorum about how the r
Progressive Medical
NOVEMBER 16, 2022
PMI LAPSYSTEM™ Electrosurgical Instruments A Sharp Tip Every Time After attending a recent laparoscopic surgeons’ conference last month […]. The post PMI LAPSYSTEM™ Electrosurgical Instruments appeared first on Progressive Medical, Inc.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
Pharmaceutical Technology
NOVEMBER 16, 2022
ElevateBio has entered a partnership with Affini-T Therapeutics to progress the latter’s engineered TCR-T therapies focused on Kirsten rat sarcoma viral oncogene homolog (KRAS), a dominant oncogenic driver mutation in solid tumours. Under the deal, the LentiPeak lentiviral vector technology platform and cell therapy production expertise of ElevateBio BaseCamp will be used by Affini-T to develop its investigational oncogenic driver programmes in the clinic.
pharmaphorum
NOVEMBER 16, 2022
With cases of drug-resistant tuberculosis on the rise again around the world, GSK has sparked hopes of a new treatment option after reporting phase 2a data with a novel antitubercular drug for the disease. The study of GSK3036656 – an inhibitor of the bacterial enzyme leucyl t-RNA synthetase (LeuRS) – showed that 14 days’ treatment with a low once-daily dose showed it was able to kill Mycobacterium tuberculosis in patients with drug-susceptible pulmonary tuberculosis , with no serious adve

PharmaVoice
NOVEMBER 16, 2022
A new CRISPR-based tool could help scientists understand and better treat cancer by revealing the complex genetic mutations that occur.

pharmaphorum
NOVEMBER 16, 2022
NICE has recommended that three COVID-19 therapies can continue to be used to treat patients – Pfizer’s Paxlovid, Roche’s RoActemra and Eli Lilly’s Olumiant – but turned down another five in draft guidance published this week. The health technology assessment (HTA) agency has turned down five other COVID-19 therapies, notably AstraZeneca’s Evusheld (tixagevimab plus cilgavimab) – which patient organisations in the UK have been clamouring for access to, as it is an alterna

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

MedCity News
NOVEMBER 16, 2022
More patients are coming back to the doctor’s office even as a physician shortage looms; here’s how savvy healthcare providers are turning to intelligent scheduling technology to optimize physician time and clinic efficiency.h.

European Pharmaceutical Review
NOVEMBER 16, 2022
Pharmaceutical company Indivior has announced it will acquire Opiant Pharmaceuticals , Inc. for $145 million in aggregate cost to support the launch of OPNT003, an opioid overdose reversal agent, over a seven-year period. According to Opiant, OPNT003 acts more quickly and lasts longer when compared with other naloxone-based opioid overdose rescue agents.
MedCity News
NOVEMBER 16, 2022
Cell and gene therapies are offering patients potentially curative treatments for a growing scope of diseases. Insurance companies are trying to figure out how to pay for them. Industry consultants speaking at the HLTH conference offered some strategies they see payers taking to these new therapies.
European Pharmaceutical Review
NOVEMBER 16, 2022
A bi-yearly publication by the Access to Medicine Foundation, the 2022 Access to Medicine Index , ranks 20 of the world’s leading research -based pharmaceutical companies on their access-to-medicine strategies for low and middle-income countries ( LMICs ), noting all 20 companies reported a late-stage R&D strategy for the first time: GSK first, Johnson & Johnson in second place and AstraZeneca in third.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
PharmaTimes
NOVEMBER 16, 2022
Findings highlight a potential new drug candidate that may delay disease progression
European Pharmaceutical Review
NOVEMBER 16, 2022
A Phase II trial showed a first-in-class, GSK-produced oral antibiotic decreased drug-susceptible pulmonary tuberculosis (TB) in all participants over 14 days, suggesting the antitubercular agent could be a simpler treatment option. A low dose of the once-daily antitubercular agent GSK3036656, demonstrated early bactericidal activity (EBA) after 14 days of treatment in Phase IIa study.

PharmaTimes
NOVEMBER 16, 2022
The therapy has the potential to treat neurological and oncological conditions
European Pharmaceutical Review
NOVEMBER 16, 2022
A Phase I study published in the Lancet Oncology , demonstrated the first implantable pump system for patients with brain cancer , effectively killed recurrent glioblastoma (GBM) tumour cells by bypassing the blood-brain barrier to continuously deliver chemotherapy through a tube inserted directly into the brain. The findings revealed the number of actively dividing glioblastoma tumour cells decreased substantially, whereas normal brain cells were not affected.

Advertisement
Clinical development organizations face a wide array of challenges when it comes to data, many of which can impact the operational effectiveness of their clinical trials. In this whitepaper, experts from Revvity Signals explore how solutions like TIBCO® Spotfire® enable better, more streamlined studies. The whitepaper also features a success story from Ambrx, a leading biopharmaceutical company, detailing how it has leveraged Spotfire to tackle data quality and collaboration challenges in clinic

Vodori
NOVEMBER 16, 2022
Having the right medical, legal, regulatory review software in place at your organization is essential for commercial success. The speed and compliance at which your team moves through the review process has a direct impact on your organization’s ability to be both proactive and reactive with your omnichannel marketing efforts.

Pharmaceutical Technology
NOVEMBER 16, 2022
Each year at the annual UN climate summits, many businesses set up pavilions at the event and have high-level meetings with policymakers. The number of companies at the COPs has been increasing each year. For some, this is a worrying sign of corporate capture. For others, it is a sign of the business community seriously engaging with the issue of climate change and telling policymakers what they need to create a stable landscape for clean investment.

pharmaphorum
NOVEMBER 16, 2022
At HLTH 2022 in Las Vegas, Editor in Chief Jonah Comstock caught up with Manny Montalvo, SVP, head of digital health and innovation at Teva Pharmaceuticals, to talk about Teva’s show news: new partnerships with Rimidi and HealthSnap to expand the deployment of the company’s Digihaler line of smart inhalers. Jonah and Manny discuss Teva’s short-term and longterm plans in the respiratory space and when a connected delivery device becomes a digital therapeutic.

Pharmaceutical Technology
NOVEMBER 16, 2022
Each year at the annual UN climate summits, many businesses set up pavilions at the event and have high-level meetings with policymakers. The number of companies at the COPs has been increasing each year. For some, this is a worrying sign of corporate capture. For others, it is a sign of the business community seriously engaging with the issue of climate change and telling policymakers what they need to create a stable landscape for clean investment.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content