The 2022 PharmaVoice 100
PharmaVoice
SEPTEMBER 6, 2022
From standout executives to R&D pioneers — this year’s class of honorees is defining the next era of leadership.

PharmaVoice
SEPTEMBER 6, 2022
From standout executives to R&D pioneers — this year’s class of honorees is defining the next era of leadership.

pharmaphorum
SEPTEMBER 6, 2022
NICE has said it is unable to recommend NHS use of AstraZeneca’s Lynparza for some patients with prostate cancer, after failing to reach an agreement on price with the drugmaker. The health technology assessment (HTA) authority said that the PARP inhibitor “is not cost-effective at its current price” as a treatment for BRCA-positive prostate cancer that has spread to other parts of the body and relapsed after prior hormonal therapy.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmaceutical Technology
SEPTEMBER 6, 2022
The US Food and Drug Administration (FDA) has awarded Orphan Drug Designation (ODD) to Avacta Group’s lead pre|CISION drug candidate, AVA6000, to treat soft tissue sarcoma. Updated using the pre|CISION technology, AVA6000 is a kind of generic chemotherapy doxorubicin. This approach aids the therapy to be activated mainly in the tumour, sparing healthy tissue from exposure.

PharmaVoice
SEPTEMBER 6, 2022
Real-world data (RWD) from clinical drug registries can and should be used throughout the drug development lifecycle. At all stages, registries ensure that assumptions about the current standard of care are accurate and evidence-based.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharma Pathway
SEPTEMBER 6, 2022
Intas Pharmaceuticals Ltd-Walk-In Drive for Manufacturing/ Packaging Department On 9th Sept’ 2022. Job Description. Company Profile: INTAS Pharmaceutical is a leading, vertically integrated global pharmaceutical formulation development, manufacturing, and marketing company. Intas is committed to challenging the unmet medical and societal needs through a comprehensive pharmaceutical value chain spanning across the world.

PharmaVoice
SEPTEMBER 6, 2022
With his unique platform, the has built a bridge between patients, physicians and researchers around the world.
Pharma Rep Focus brings together the best content for pharma rep professionals from the widest variety of industry thought leaders.

PharmaVoice
SEPTEMBER 6, 2022
The CMO at Kura Oncology, a clinical-stage biopharma company, is charting a new course to deliver on the promise of precision medicine in oncology.

MedReps
SEPTEMBER 6, 2022
It’s not unusual for a medical sales representative, regardless of their product niche, to hear price negotiations from their clients. After all, everyone wants the best price, even if that involves taking a mere 5% off of the cost. Even the largest hospitals and clinics have budgets and it’s up to your clients to adhere to them. With that said, how do you address the price issue when it comes up?

pharmaphorum
SEPTEMBER 6, 2022
Pleco Therapeutics has closed a first-round financing, raising just over €17 million that it says will fund the development of its lead therapy for acute myeloid leukaemia (AML) through to regulatory filings in the US and EU. The Nijmegen, Netherlands-based biotech was founded in 2018 to develop therapies designed to change the balance of protein expression within the tumour ‘microenvironment’, which can defend cancers against the immune system, and is increasingly becoming a source

PharmaVoice
SEPTEMBER 6, 2022
As a patient advocate, The Spark Global’s owner is transforming how pharma companies approach patient centricity.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

pharmaphorum
SEPTEMBER 6, 2022
New UK Prime Minister Liz Truss needs to take immediate action to prevent the NHS spiralling out of control this winter, according to groups representing healthcare workers. Truss takes power as the NHS is in “its worst state in living memory,” according to NHS Confederation chief executive Matthew Taylor, who says it is vital that the health service becomes a “top three” priority for the new administration as it grapples with an estimated real-terms funding cut of £9.4 b

PharmaTimes
SEPTEMBER 6, 2022
PhoreMost will use Siteseeker platform to identify novel targets for Roche’s discovery programmes

pharmaphorum
SEPTEMBER 6, 2022
The healthcare sector has a “moral imperative” to help people navigate the self-care industry, says pharma giant. Recent years have seen an explosion in the “self-care” industry, a sector that was worth an estimated $4.9 billion in 2019. But in a world of fake news and influencer-led trends, it’s the healthcare sector’s responsibility to ensure people have the knowledge they need to sort the wheat from the chaff.

Pharmaceutical Technology
SEPTEMBER 6, 2022
The pharmaceutical supply chain is a global and complex network that includes a wide range of stakeholders such as pharmaceutical manufacturers, wholesale distributors, and pharmacy benefit managers (PBM), to ensure efficient and timely delivery of medications to the patients. Hospitals, clinics, pharmacies, wholesalers of medical products and other health centres are catered by pharmaceutical logistics that include handling activities such as procurement, warehousing, and inventory management,

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

PharmaTimes
SEPTEMBER 6, 2022
Group consists of nine ventures plus corporate-sponsored innovation projects from Novo Nordisk

PharmaTech
SEPTEMBER 6, 2022
How Pfizer’s external-facing research and development scientists uncover the most promising emerging therapy concepts and ideas in the pharmaceutical landscape.

Pharma Pathway
SEPTEMBER 6, 2022
Aculife Healthcare Pvt. Ltd-Walk-In Interview for Freshers for Apprentice On 15th Sept’ 2022. Job Description. Walk-In Interview for Freshers for Apprentice On 15th Sept’ 2022 @ Aculife Healthcare Pvt. Ltd. Job Role: Apprentice Trainee. B.Sc/ M.Sc. B.Pharm/ M.Pharm. B.Com/ M.Com/ BA/ MA. Fitter Electrical / Turner. Wireman/ Welder/ COPA.

Scott’s Directories
SEPTEMBER 6, 2022
Many business owners are not aware that bad data can affect their revenue. According to statistics from Experian Data Quality, an average company loses about 12% of its revenue because of bad data. Bad data can cause business organizations to pay heavy fines. For example, many companies have been fined for sending text messages to mobile numbers that were reassigned to a new user.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

Pharmaceutical Technology
SEPTEMBER 6, 2022
Business optimism improved in August 2022 compared to July 2022, according to an ongoing poll by Verdict, despite businesses expressing concern over inflation, particularly in raw materials and energy costs. Verdict has been conducting the poll to study the trends in business optimism during COVID-19 as reflected by the views of companies on their future growth prospects amid the pandemic.
Legacy MEDSearch
SEPTEMBER 6, 2022
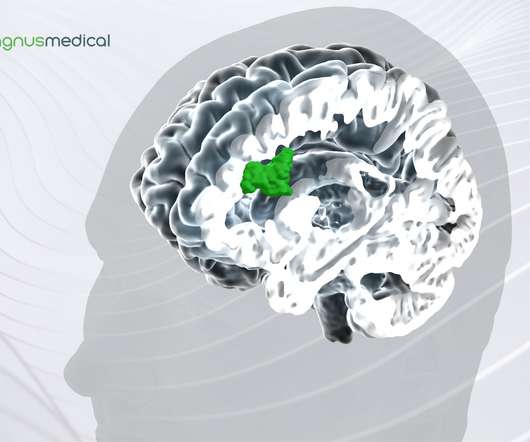
Magnus Medical, Inc. , a medical device company and developer of brain stimulation technology for treatment of neuropsychiatric disorders, announced it received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for the SAINT TM Neuromodulation System for the treatment of major depressive disorder (MDD) in adults who have failed to achieve satisfactory improvement from prior antidepressant medications in the current episode.

Scott Burrows
SEPTEMBER 6, 2022
Safety is not about “Hybrid” The business world, it seems, is obsessed with the hybrid workplace. As a motivational safety speaker who once “worked with his hands,” I see the obsession with the office workplace as perhaps a bit classist.
pharmaphorum
SEPTEMBER 6, 2022
The EU has blocked the merger of Illumina and Grail, saying the deal would stifle innovation in blood-based diagnostics for cancer diagnostics, just days after a bid by the US authorities to do likewise was blocked in the courts. The European Commission said in a statement this afternoon that Illumina had not provided enough assurances to address its concerns that the $7.1 billion merger would squeeze other players in the liquid biopsy category out of the market.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

PharmaVoice
SEPTEMBER 6, 2022
The chief medical officer at physIQ is working at the intersection of healthcare, clinical trials and technology to deliver better, faster and more accurate treatments to patients around the globe.

Pharmaceutical Technology
SEPTEMBER 6, 2022
The UK’s pharma industry registered a 3.3% rise in IT hiring activity in August 2022 when compared with the previous month, according to GlobalData’s Job Analytics database. The industry’s overall hiring is decreased by 1.6% in August 2022 when compared with July 2022. Out of the pharma industry’s total hiring activity, IT jobs claimed a 4.27% share in August 2022, recording an increase of 0.13% over the last three-month average share.

pharmaphorum
SEPTEMBER 6, 2022
Pharmacy chain CVS has stepped up its drive into the wider healthcare market with an agreement to buy at-home care and technology provider Signify Health for around $8 billion – reportedly besting Amazon in a bidding war for the group. CVS is paying $30.50 per share for Texas-based Signify, which operates a network of more than 10,000 clinicians across all 50 states, delivering care to patients with the help of a digital health and analytics platform to Medicare Advantage and other federal manag

Pharmaceutical Technology
SEPTEMBER 6, 2022
Switzerland’s pharma industry registered a 2.3% drop in IT hiring activity in August 2022 when compared with the previous month, according to GlobalData’s Job Analytics database. The industry’s overall hiring is decreased by 1.62% in August 2022 when compared with July 2022. Out of the pharma industry’s total hiring activity, IT jobs claimed a 2.74% share in August 2022, recording a decrease of 0.53% over the last three-month average share.

Advertisement
Clinical development organizations face a wide array of challenges when it comes to data, many of which can impact the operational effectiveness of their clinical trials. In this whitepaper, experts from Revvity Signals explore how solutions like TIBCO® Spotfire® enable better, more streamlined studies. The whitepaper also features a success story from Ambrx, a leading biopharmaceutical company, detailing how it has leveraged Spotfire to tackle data quality and collaboration challenges in clinic

PharmaVoice
SEPTEMBER 6, 2022
The longtime regulatory pro has used innovative strategies to deliver emerging oncology therapies to patients from her position as head of regulatory affairs at Stemline.

Pharmaceutical Technology
SEPTEMBER 6, 2022
The US’s pharma industry registered a 1.4% drop in IT hiring activity in August 2022 when compared with the previous month, according to GlobalData’s Job Analytics database. The industry’s overall hiring increased by 2.81% in August 2022 when compared with July 2022. Out of the pharma industry’s total hiring activity, IT jobs claimed a 1.84% share in August 2022, recording a decrease of 0.05% over the last three-month average share.

PharmaVoice
SEPTEMBER 6, 2022
The COO has launched two jewels in the biotech world that complement each other while serving broader innovation goals throughout the industry.

Pharmaceutical Technology
SEPTEMBER 6, 2022
UK-based company AstraZeneca’s IT hiring declined 5.4% in August 2022 when compared with the previous month, according to GlobalData’s Job Analytics database. The company’s overall hiring activity declined by 8.49% in August 2022 when compared with July 2022. IT jobs claimed an 8.47% share in the company’s total hiring activity in August 2022, and recorded a 0.24% growth over the last three-month average share.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content