Embracing Personalization in Biopharma Patient Support Programs
Fierce Pharma
APRIL 18, 2023
Embracing Personalization in Biopharma Patient Support Programs mteefey Tue, 04/18/2023 - 14:48
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Fierce Pharma
APRIL 18, 2023
Embracing Personalization in Biopharma Patient Support Programs mteefey Tue, 04/18/2023 - 14:48

Fierce Pharma
FEBRUARY 27, 2024
Delivering on these heightened expectations has fueled the urgency to invest in patient engagement and support. A seismic shift has occurred in the healthcare landscape.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PM360
APRIL 22, 2024
This is especially relevant in biopharmaceutical patient engagement, where regulatory infractions can have existential consequences. The Unique Challenges of Patient/Pharma Interaction Patient engagement has become a critical component of medical marketing.

pharmaphorum
DECEMBER 18, 2022
Science has taken a vertical growth trajectory, yet such innovations are, at best, slow to reach the patients in need. We believe that innovation in patient access models is the necessary catalyst to aid patients in starting on and staying on the therapies they need to improve their lives.

PM360
FEBRUARY 1, 2024
Embarking on the journey from product development to market success in the biopharma industry is no small feat. According to a McKinsey report , a startling 40% of biopharma products fail to meet their sales forecasts within the initial two years, prompting a critical reassessment of strategies for product launches.

European Pharmaceutical Review
SEPTEMBER 4, 2023
The biopharma company will be required to seek prior approval before acquiring related products. BIO stated that the settlement will enable the FTC to protect competition, while facilitating rare disease patients to promptly benefit from the deal’s pro-competitive advantages. Divest to invest: the new normal in biopharma?

European Pharmaceutical Review
AUGUST 14, 2023
The goal of such therapies is to free patients from constant disease management by sensing blood glucose levels, restoring insulin production and releasing it over the long term. “By The post Lilly completes biopharma acquisitions appeared first on European Pharmaceutical Review. Both companies have been working together since 2018.

European Pharmaceutical Review
DECEMBER 20, 2023
1 The changes form part of the 2020 Pharmaceutical Strategy for Europe will impact pharma and biopharma companies operating in Europe. The second is an adjustment of unmet need categorisation required for RDP extension; unmet need definition now encompasses ‘meaningful’ impact patient outcomes (notably including quality of life).

MedCity News
OCTOBER 27, 2023
Technology is transforming how clinical trials are run. A panel during MedCity News’ INVEST Digital Health conference discussed the tech trends shaping the design and execution of drug studies.

Pharmaceutical Technology
OCTOBER 4, 2022
Treatments to enhance the lives of debilitating neurological and neuropsychiatric disease patients are part of Biohaven’s portfolio. Combined with Pfizer’s global reach, this acquisition increases our potential to bring new treatment options to patients with migraine – a disease which affects over one billion people worldwide.”.

Fierce Pharma
SEPTEMBER 19, 2023
With the right infrastructure, intelligent tools stand to not only process data but also improve clinical decision-making—helping find more patients for biopharma companies, and faster, too. Especially with the proliferation of AI in other industries and chatGPT.
Pharmaceutical Technology
MAY 26, 2023
HanAll Biopharma and Daewoong Pharmaceutical have entered into a co-development deal with NurrOn Pharmaceuticals to develop therapy for Parkinson’s disease (PD) and other neurodegenerative disorders. By activating Nurr1, ATH-399A slows disease progression in PD patients. They will help to progress ATH-399A towards a Phase I trial.

Pharmaceutical Technology
JANUARY 24, 2023
BioCryst Pharmaceuticals is collaborating with Swixx BioPharma for Orladeyo (berotralstat) commercialisation in Central and Eastern Europe (CEE). Under the deal, Swixx BioPharma will handle the commercialisation activities of oral therapy in 15 CEE markets.

Pharmaceutical Technology
SEPTEMBER 8, 2022
We have focused on PD-1-IL-2 as a biology that has great potential for benefiting patients.”. The post Roche to acquire biopharma firm Good Therapeutics for $250m appeared first on Pharmaceutical Technology.

Pharmaceutical Technology
SEPTEMBER 2, 2022
Forma focuses on researching, developing and marketing new therapies for patients with sickle cell disease (SCD), rare hematologic diseases and cancers. An oral, investigational, once-a-day, selective pyruvate kinase-R (PKR) activator, etavopivat is being developed to boost anaemia and the health of red blood cells in SCD patients.
Pharmaceutical Technology
MAY 10, 2023
Swedish Orphan Biovitrum (Sobi) has entered into a definitive agreement to buy biopharmaceutical company CTI BioPharma in an all-cash deal valued at $1.7bn. Vonjo has received the US Food and Drug Administration’s approval to treat myelofibrosis in adult patients with a platelet count below 50 x109/L.
MedCity News
APRIL 23, 2024
ImmunityBio therapy Anktiva is now FDA approved for treating patients with non-muscle invasive bladder cancer. The post FDA Approves Immunotherapy That Can Spare Bladder Cancer Patients From Radical Surgery appeared first on MedCity News. The novel immunotherapy works by sparking activity from three types of immune cells.

Pharmaceutical Technology
AUGUST 5, 2022
“We are excited to join in the Tavneos launch and help many more patients with this serious and sometimes life-threatening disease for which there remains significant unmet medical need.”. The post Amgen to acquire biopharma firm ChemoCentryx for $3.7bn appeared first on Pharmaceutical Technology.

Pharmaceutical Technology
JUNE 2, 2023
Rentschler Biopharma has collaborated with Ikarovec for expediting the new gene therapies to treat ophthalmic disease. Under the deal, Rentschler Biopharma’s ATMP facility in Stevenage, UK, will undertake the bioprocess development of adeno-associated virus (AAV) material.

pharmaphorum
JUNE 27, 2022
It is also approved for patients aged 16 years and older with metastatic or locally advanced epithelioid sarcoma not eligible for complete surgical removal, and is in phase 3 testing as a combination therapy with rituximab and lenalidomide as a second-line therapy for FL. .

Pharmaceutical Technology
OCTOBER 12, 2022
Biotech start-up Toregem BioPharma has signed a memorandum of understanding (MoU) with WuXi Biologics to develop the monoclonal antibody, TRG035. We look forward to bringing this tooth regeneration drug to the global market, treating patients across the world.”. TRG035’s Phase I clinical study is scheduled to begin in early 2024.

Pharmaceutical Commerce
DECEMBER 6, 2023
Matt Wolf, president of biopharma services, Cencora, discusses the importance of partnership in helping to combat prescription drug abandonment and non-adherence.

Clarivate
MARCH 8, 2023
In recent years, there has been an increased focus on real world evidence to advance medical science and improve patient quality of life. Biopharma companies are investing in the use of real world data (RWD) to accelerate innovation. The treatment of patients experiencing acute disease is quite dynamic.
Pharmaceutical Technology
JUNE 24, 2022
invoX Pharma has signed a definitive agreement for the acquisition of all issued and outstanding shares of clinical-stage biopharma firm F-star Therapeutics, in a deal valued at nearly $161m or $7.12 The post invoX Pharma agrees to acquire biopharma firm F-star Therapeutics for $161m appeared first on Pharmaceutical Technology.

MedCity News
MARCH 6, 2024
This case, about the claim that Merck failed to warn patients about the risk of typical femoral fracture, reaches past this suit to potentially change pharmaceutical companies’ relationship with the FDA.
pharmaphorum
AUGUST 5, 2022
Over the past 15 years, more than 60% of asset growth among the top 30 biopharma companies came from acquisitions. The pace of innovation in biopharma has rocketed over the last two years, with vaccines, new antivirals, and therapies delivered at an unprecedented pace. About the Author.

pharmaphorum
APRIL 4, 2024
The company said that it was stopping access to the drug for new patients immediately, while those already taking it would be able to continue on it via a free drug programme, after consultation with their doctors – a move that has garnered praise from across the biopharma industry.
MedCity News
APRIL 8, 2024
The expanded FDA approvals makes these cell therapies more accessible to more patients, broadening the market for both products. The post Cell Therapy Now Set to Reach More Patients With Expanded FDA Nod for 2 Multiple Myeloma Drugs appeared first on MedCity News.

PharmExec
APRIL 7, 2023
Biopharma companies can foster patient-centered innovation by focusing on unmet needs along each stage of the patient journey while collaborating with key stakeholders directly or indirectly responsible for disease management.

Pharmaceutical Technology
SEPTEMBER 20, 2022
The company intends to commence a randomised, controlled Phase II proof-of-concept clinical trial of VS-01, enrolling 60 ACLF patients in the fourth quarter of the year. The post Genfit agrees to acquire biopharma firm Versantis for $109m appeared first on Pharmaceutical Technology.

MedCity News
DECEMBER 19, 2022
Historically, clinical trials have not prioritized the experiences of patients, who are required to frequently return on-site and engage with unfamiliar medical technology. They are often left in the dark regarding the data and health outcomes being collected through their participation.

MedCity News
FEBRUARY 20, 2024
The FDA clinical hold follows a patient death from liver failure that may be associated with zelnecirnon, an experimental Rapt Therapeutics drug. The biotech notes that no other patient has experienced liver problems and this safety signal has not been seen in any other tests of the molecule.
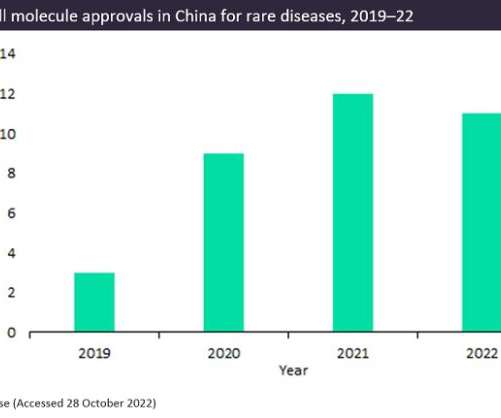
Pharmaceutical Technology
NOVEMBER 2, 2022
However, over the same period, approvals for innovator small molecules treating rare diseases rose by 22%, giving hope to rare disease patients. The post China’s biopharma industry: the small molecule renaissance in rare diseases appeared first on Pharmaceutical Technology.

MedCity News
MAY 19, 2023
Companies presenting their technology at the conference seek to support a diverse patient population spanning conditions such as inflammatory bowel disease, food allergies, and cancer. Cell, gene therapy and immunotherapies also form some of their treatment approaches in development.

Clarivate
JUNE 29, 2023
We’re mindful of some challenges that life science and healthcare organizations must work through before this technology is mature enough for use in critical business decisions that may impact patient health. Our customers entrust our products and services to help them improve patient health, and we will not jeopardize that mission.

MedCity News
SEPTEMBER 12, 2022
As the biopharma space gets more competitive and the days of blockbuster drugs seem further in the rearview, biopharma companies are investing in digital to find new and innovative ways to differentiate themselves.

PharmaVoice
OCTOBER 16, 2023
Biopharma leaders pounding away at rare diseases so patients with few other options can have healthier lives.

MedCity News
OCTOBER 16, 2023
With increased access to cutting-edge imaging, improved treatment options, greater personalization of treatment, reduced radiation exposure, and potential for future advances, patients can expect to receive more advanced medical care tailored to their specific needs through radiopharmaceuticals.

European Pharmaceutical Review
MAY 12, 2023
Swedish Orphan Biovitrum AB (Sobi ® ) has agreed to acquire CTI BioPharma for $1.7 There is a large unmet medical need within myelofibrosis, in particular for patients suffering from thrombocytopenia who are inadequately treated by existing medicines.
Pharmaceutical Technology
AUGUST 9, 2022
“The deep market knowledge and scientific and clinical capabilities we have built over three decades in rare haematology will enable us to accelerate innovation for the sickle cell disease community and bring these treatments to patients as quickly as possible.”.

MedCity News
JULY 20, 2022
By adding adherence and connectivity capabilities to home-based solutions, clinicians can maintain open lines of communication with patients, helping patients better care for themselves from the comfort of their homes.

PM360
SEPTEMBER 15, 2022
The term “patient centricity” has been around for over a decade, and since its first utterance the buzzword has been praised, dissected, criticized, and everything in between. The concept has always been worthwhile, but people within and outside of the industry have wondered whether the industry was truly becoming more patient centric?

MedCity News
FEBRUARY 26, 2023
Wearable patient devices can provide healthcare organizations with more varied data, expand options for clinical trials, and make patients more active participants in their own care.

MedCity News
NOVEMBER 1, 2023
Investing in comprehensive patient concierge services keeps trial administrators on track by removing the burden of logistical support and improving the overall participant experience, making dropouts and trial delays much less likely.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content