Zydus Pharmaceuticals (USA) Inc. Profile
Pharmacy Times
AUGUST 22, 2023
Zydus Pharmaceuticals (USA) Inc, has come a long way since its first commercial launch in August 2005.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
 2005 Related Topics
2005 Related Topics 
Pharmacy Times
AUGUST 22, 2023
Zydus Pharmaceuticals (USA) Inc, has come a long way since its first commercial launch in August 2005.

Pharma Pathway
DECEMBER 28, 2022
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Macleods Pharmaceuticals, Indian’s fastest growing company was established in 1986.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharma Pathway
FEBRUARY 10, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
FEBRUARY 1, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
FEBRUARY 26, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
FEBRUARY 4, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. .- Walk-In Interviews for Production/ Quality Control On 5th Feb’ 2023 Job Description Macleods Pharmaceuticals, Indian’s fastest growing company was established in 1986.

Pharma Pathway
JANUARY 7, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Macleods Pharmaceuticals, Indian’s fastest growing company was established in 1986.

Pharma Pathway
MARCH 13, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
JANUARY 1, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Macleods Pharmaceuticals, Indian’s fastest growing company was established in 1986.

Pharma Pathway
FEBRUARY 11, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
MARCH 2, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
MARCH 4, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
MARCH 2, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

World of DTC Marketing
JANUARY 3, 2022
spent $190 billion on obesity-related health care expenses in 2005-double previous forecast. (1) Compared with those with average weight, adults with obesity in the United States experienced higher annual medical care costs by $2,505 or 100%, with prices increasing significantly with the class of obesity. By one estimate, the U.S.

Pharma Pathway
FEBRUARY 1, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Ranked amongst the top 10 pharma companies with a strength of 20000 employees, operating in 140+ countries.

Pharma Pathway
JANUARY 5, 2023
Our bioequivalence centre was established in 2005 and is responsible for conducting studies for filing product registrations with various regulatory authorities. Macleods Pharmaceuticals, Indian’s fastest growing company was established in 1986.
Medical Device Success
OCTOBER 10, 2021
Over 30 years ago, Paula began her career in the medical device industry and in 2005, founded Legacy MedSearch and turned it into one of the most highly sought after MedTech search firms in the USA. Reading time: 2 – 4 minutes. More change in the last 18 months than in the past 30 years.”.

PM360
MARCH 22, 2024
A study from The National Cancer Institute revealed that the number of women who were unaware they should get these screenings went up from 45% in 2005 to 55% in 2019. The number of women who said they did not get a recommendation from a healthcare professional to get these screenings also doubled, from 6% in 2005 to 12% in 2019.

PM360
MARCH 24, 2023
Within the past 40 years, 202 new awareness days were introduced to Congress—71% of these were introduced after 2005. We’re seeing more and more awareness days, weeks, and months emerge in large part due to the rise of social media. This is right around the time social media was really taking off.
pharmaphorum
OCTOBER 7, 2022
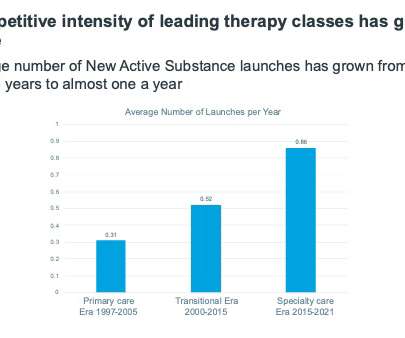
Starting with the primary care era of 1997-2005, when key launches were primary care blockbusters, such as Losec, Diovan, and Lipitor, IQVIA took five key therapy classes from that era that consistently appeared in the top 10 therapy area by sales for the time period of the era.

European Pharmaceutical Review
OCTOBER 2, 2023
Through [their] discoveries, Karikó and Weissman opened up opportunity for the pharmaceutical industry to advance mRNA technology” These data were published in 2005, fifteen years before the COVID-19 pandemic, of which mRNA vaccines were instrumental to controlling.

Copyright Clearance Center
SEPTEMBER 1, 2022
Foster has been a member of CCC’s Board since 2005, previously holding the position of Audit Committee Chair, and currently serves on the Executive Committee. Foster succeeds Jack Hoeft, who served as Chairman for over 20 years.

Pharma Marketing Network
MAY 3, 2023
As such, it is important for the industry to carefully consider the effects of these advertisements and to take steps to ensure that they are promoting the right messages in the right ways. References: 1. Powerful Medicines: The Benefits, Risks, and Costs of Prescription Drugs. Knopf Doubleday Publishing Group. Goozner, M. Steinman, M.

Clarivate
NOVEMBER 1, 2021
These include Handbook of Quantitative Science and Technology Research (2004), Citation Analysis in Research Evaluation (2005), Applied Evaluative Informetrics (2017), Handbook of Science and Technology Indicators (edited with W. His books became the cornerstone of bibliometrics education. Glänzel, U. Schmoch and M. Thelwall) in 2018.

Clarivate
MAY 8, 2023
Artios Pharma CEO Niall Martin recalled the sale of KuDOS Pharmaceuticals to AstraZeneca for $210m in late 2005 and advised that there will always be a question mark about the right time to sell when there is pressure in the system to sell assets, which is mirrored in today’s biotech environment.

World of DTC Marketing
JUNE 5, 2021
From 2005 to 2014, most cancers associated with overweight and obesity increased in the United States, while cancers associated with other factors decreased. Breast (in women who have gone through menopause). Colon and rectum. Gallbladder. Upper stomach. Meningioma (a type of brain cancer). Multiple myeloma.

PM360
AUGUST 9, 2022
According to a 2021 study by Statista, 40% of prescriptions written in 2005 were for brand-name medications. By 2020, that percentage plummeted to just 8%. 1 How is this possible, given that more than 20,000 prescription drugs are currently in circulation in the U.S.

pharmaphorum
NOVEMBER 7, 2022
Since Mitch helped to found Aerie as a Duke University spinout in 2005, Aerie has filed 5 INDs and 2 NDAs, with its first FDA approval coming in 2017 and Najam (Naj) Sharif, PhD, DSc (Southampton University, UK) has >34-years’ pharmaceutical drug discovery research/development experience covering neuroscience and ophthalmology.
Pharmaceutical Technology
FEBRUARY 14, 2023
In 2005, the FDA approved Bristol-Myers Squibb's Abraxane, a PX albumin-bound nanoparticle formulation with 130nm particles, for treating metastatic breast cancer. Bristol-Myers Squibb is the leading patent filer in nanoparticles for drug delivery.

Medico Reach
AUGUST 1, 2022
The whole Sprout healthcare technology team broke out into NLV in 2005. As a spin-off of the Sprout Group, NLVP has been participating in biopharma since 2005. New Leaf Venture Partners (NLVP) invested in biopharmaceuticals during their growth and venture phases.

pharmaphorum
NOVEMBER 25, 2022
David James has a long history within global product forecasting and consulting and co-launched J+D in 2005. About the author. He became CEO in 2011, as the company moved from purely consulting to developing forecast models, and has continued to evolve the business, using innovative technology to drive efficiencies for clients’ processes.

Legacy MEDSearch
AUGUST 31, 2023
More information: Expert insights on how to navigate the new mandatory A2P 10DLC industry requirements FCC Fact Sheet on Text Messaging Text Message Revenue Growth Since 2005 Text Carriers Announce New Requirements Our team is working hard to deliver the best experience to our candidates and hiring managers, and we respect anyone’s option (..)

pharmaphorum
OCTOBER 6, 2022
He served as Director of the Lowe Family Genomics Core at the Johns Hopkins University from 2005-15. He trained in Molecular Biology and earned a doctorate in Genetics from George Washington University, followed by a postdoctoral fellowship at the National Institute of Aging in the Microarray Core.

pharmaphorum
JANUARY 27, 2023
He previously held regional leadership roles at Sandoz between 2005 and 2010. Saynor has a wealth of experience in the pharmaceutical industry, including at both innovation-driven companies and generics makers.

European Pharmaceutical Review
MARCH 1, 2023
PharmTech 2005 Apr 1. J Food Science 2005; 70(7): e437-e442 [[link] 5. Measurement of density variations in tablets using X-ray computed tomography. Int J Pharm 2004; 271(1-2): 215–24 [[link] 15129988] 2. Hancock BC, Mullarney MP. X-ray Microtomography of Solid Dosage Forms. Sovány T, Kása P, Vakli K, Pintye-Hódi K. X-Ray Spectrom.

European Pharmaceutical Review
OCTOBER 19, 2022
FDA highlighted that over three quarters of new patents in the Orange Book between 2005 and 2015 were assigned to existing drugs. 10 There is a fear that such practices discourage innovative approaches, impacting on patient welfare. Finally, product hopping is another tactic for hindering generic entry into the market.

Clarivate
NOVEMBER 17, 2021
After a successful time there, where I established an international reputation and worked with some outstanding colleagues, I moved back to the United States in 2005 and then onto my current position at the University of Sydney in 2012.

European Pharmaceutical Review
JULY 13, 2023
2005 Dec;146(7):913-5. Expert Review of Clinical Pharmacology. 2023 Mar 4;16(3):257-66. Mechoulam R. Plant cannabinoids: a neglected pharmacological treasure trove. British journal of pharmacology. Hay M, Thomas DW, Craighead JL, et al. Clinical development success rates for investigational drugs. Nature biotechnology. 2014 Jan;32(1):40-51.

Clarivate
NOVEMBER 14, 2023
The first GLP-1 RA used for diabetes (short-acting exenatide) became available in 2005 (US)/2006 (Europe) and required twice-daily injection. Since then, two GLP‑1 RAs requiring only once-daily injection have become available, followed by three which require only once-weekly injection (Figure 1). [7]
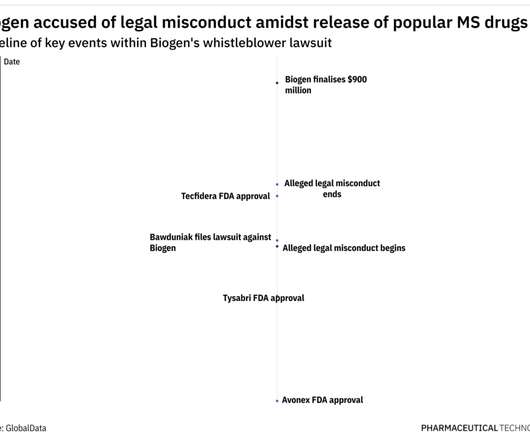
Pharmaceutical Technology
OCTOBER 5, 2022
In 2005, Biogen withdrew Tysabri from the market following a clinical trial that resulted in two participants contracting progressive multifocal leukoencephalopathy (PML). It is also important to ask whether doctors are being paid fair market value and if they are providing the services for which they’re being compensated, he adds.

European Pharmaceutical Review
OCTOBER 30, 2023
Graham originally qualified as a solicitor in London in 2005, before moving to co-found technology and software company MET, which was subsequently sold to a group of private investors in 2008.

pharmaphorum
OCTOBER 31, 2022
David James has a long history within global product forecasting and consulting and co-launched J+D Forecasting in 2005. By defining the remit, you can begin to identify whether features within the model are necessary additions or ‘nice-to-haves’ to keep it simple. About the author.

European Pharmaceutical Review
APRIL 21, 2023
Giving environmental risk assessments teeth Since 2005, the authorisation of a human pharmaceutical product requires an environmental risk assessment (ERA). In addition, the number of active substances detected in the environment has been increasing with unknown consequences for ecosystems and the accumulation of substances in food chains.

pharmaphorum
AUGUST 17, 2022
They are a stripped-down version with 29 indicators; the 2005 PICTF had 46. Indicators from the 2000s were captured in the Pharmaceutical Industry Competitiveness Task Force (PICTF) Indicators. The July 2022 LSCIs are the eighth version.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content